Evaluate
* Answer each question in the series below to view Advice.
What is the SEVERITY of Patient's MR?
Which of the Following did Diagnostic Testing Indicate?
Select All Criteria Typical of Severe MR That Apply to Your Patient:
What is the ETIOLOGY of the Patient's MR?
Large Crescentic Orifice?
Select All Criteria Typical of Mild MR That Apply to Your Patient:
Hidden Severity
Hidden Etiology
Select Quantitative Grade That Best Describes Your Patient:
Is Flail Leaflet or Tethering Present?
Candidate for surgery?
Select Leaflet Morphology
LVEF < 50%?
Is revascularization indicated?
Is CABG preferred or is there a need for other surgery?
After initiating or intensifying GDMT, does the patient have ongoing HF symtpoms?
Candidate for CRT?
Likelihood of successful repair > 95% AND Peri-op risk < 1%?
Select the Carpentier Classification of Leaflet Motion
Select Symptom Status
Are Any of the Following Additional Characteristics Present?
• Ejection Fraction < 0.6
• Left Ventricular End-Systolic Diameter > 4.0 cm
• Serial Increase in LV Size or Decrease in LV Function Over Time
• Recent-onset Atrial Fibrillation
• Pulmonary Artery Systolic Pressure > 50 mmHg
Advice
Inputs
Resources
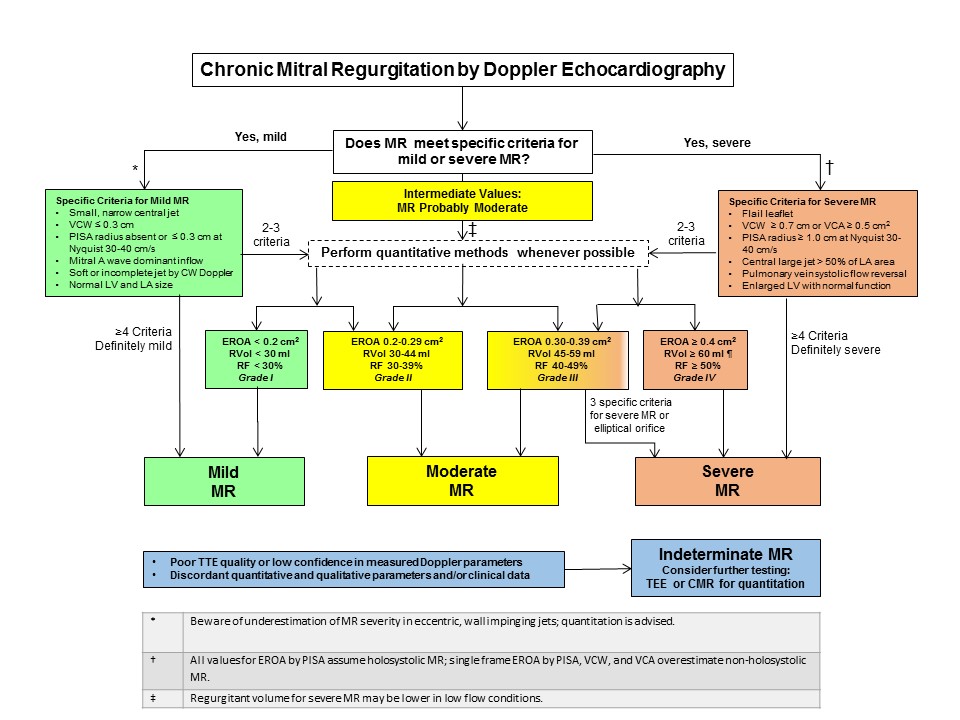
Decision Tree for Assessing Severity of Chronic MR by TTE
Strengths and Limitations of Common ECHO Parameters
Pathoanatomically Directed Surgical Techniques for MR
Feasibility of Surgical Mitral Valve Repair
Eligibility for Transcatheter Mitral Valve Intervention
Feasibility of Transcatheter Edge-to-Edge Clip Repair
Clinician Tools
Patient Tools and Resources
Other Mobile Tools
References
Bonow RO, O’Gara PT, Adams DH, Badhwar V, Bavaria JE, Elmariah S, Hung JW, Lindenfeld J, Morris AA, Satpathy R, Whisenant B, Woo YJ
2020 Focused Update of the 2017 ACC Expert Consensus Decision Pathway on the Management of Mitral Regurgitation
Journal of the American College of Cardiology (2020), doi: https:// doi.org/10.1016/j.jacc.2020.02.005
Carpentier Atrial View Image: Castillo JG, Solís J, González-Pinto Á, Adams DH.
Surgical Echocardiography of the Mitral Valve.
Revista Española de Cardiología (English Edition) 2011;64:1169-1181.
O’Gara PT, Grayburn PA, Badhwar V, Afonso LC, Carroll JD, Elmariah S, Kithcart AP, Nishimura RA, Ryan T, Schwartz A, Stevenson LW.
2017 ACC Expert Consensus Decision Pathway on the Management of Mitral Regurgitation: A Report of the American College of Cardiology Task Force on Expert Consensus Decision Pathway.
J Am Coll Cardiol. 2017 Nov, 70 (19) 2421–2449.
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP, Fleisher LA, Jneid H, Mack MJ, McLeod CJ, O’Gara PT, Rigolin VH, Sundt TM, Thompson A.
2017 AHA/ACC Focused Update of the 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines.
J AM Coll Cardiol 2017: DOI: 10.1016/j.jacc.2017.03.011
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP, Guyton RA, O’Gara PT, Ruiz CE, Skubas NJ, Sorajja P, Sundt TM, Thomas JD.
2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines.
J AM Coll Cardiol 2014:63;57-185
Nishimura RA, O’Gara PT, Bavaria JE, Brindis RG, Carroll JD, Kavinsky CJ, Lindman BR, Linderbaum JA, Little SH, Mack MJ, Mauri L, Miranda WR, Shahian DM, Sundt TM 3rd.
2019 AATS/ACC/ASE/SCAI/STS expert consensus systems of care document: a proposal to optimize care for patients with valvular heart disease: a joint report of the American Association for Thoracic Surgery, American College of Cardiology, American Society of Echocardiography, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons.
J Am Coll Cardiol 2019;73:2609–35
Terms of Service
ManageMR Terms of Service and License Agreement
This Terms of Service and License Agreement constitutes a legal agreement (collectively, the "Agreement") between the American College of Cardiology Foundation ("ACCF") and You and your agents ("You") for the use of the ManageMR App (the "Product"), whether You use the mobile application version of the Product or the web version of the Product. The Product allows You to access certain content included in the Product ("Content") relating to referral and treatment advice for patients with mitral regurgitation.
By using the Product, You accept and agree to be bound by all of the terms and conditions set forth in this Agreement. If You do not wish to accept the terms and conditions of this Agreement, You may not proceed to use the Product.
ACCF may change the terms of this Agreement from time to time without further notice directly to You. When the terms are changed, ACCF will post a general conspicuous notice. If You do not agree with the revised terms, please discontinue use of the Product immediately. Your continued use of the Product following such notice constitutes your acceptance of and agreement to be bound by any revised terms of the Agreement. This Agreement expressly incorporates by reference and includes rules or disclaimers that may be posted and updated within the Product or communicated to You from time to time.
Term and Termination
ACCF may terminate your access and/or the Product at any time. You agree that any termination of your access to the Product shall not result in any liability or other obligation of ACCF to You, or any third party in connection with such termination.
Intellectual Property Rights
This Product, including the information, text, graphics, images, audio and video files, trademarks and other materials that may be contained therein (collectively "Content"), is owned by ACCF and/or its suppliers and is protected by patents, copyrights, trademarks, and other proprietary rights. Except as specifically provided in this Agreement, your use of the Product shall be governed and constrained by applicable patent, copyright, trademark and other intellectual property laws.
ACCF grants You a limited, nonexclusive, nontransferable, revocable license to utilize and access the Product for your noncommercial, personal use according to the terms and conditions in this Agreement. You may not modify, publish, transmit, participate in the transfer or sale of, reproduce, create derivative works from, distribute, perform, display, incorporate into another website, or in any other way exploit the Service and/or any of the Content, in whole or in part. Except as expressly granted by this Agreement, You acquire no right, title or interest in the Product or the Content or other data or materials incorporated in the Product. ACCF, ACC or affiliates or licensors thereof shall retain all right, title and interest in the Product and Content.
Limitation of Liability
TO THE FULLEST EXTENT ALLOWED BY APPLICABLE LAW, ACCF HEREBY DISCLAIMS, AND IN NO EVENT SHALL ACCF OR ANY PARTY INVOLVED IN CREATING OR PRODUCING THE PRODUCT BE LIABLE FOR, ANY DIRECT, INDIRECT, INCIDENTAL, SPECIAL, CONSEQUENTIAL, OR EXEMPLARY DAMAGES, INCLUDING WITHOUT LIMITATION, DAMAGES FOR LOSS OF PROFITS, GOODWILL, USE, DATA LOSS, OR OTHER LOSSES, WHETHER IN AN ACTION OF CONTRACT, NEGLIGENCE OR OTHER TORTIOUS ACTION, EVEN IF ACCF HAS BEEN ADVISED OF THE POSSIBILITY OF SUCH DAMAGES, RESULTING FROM: (i) THE USE OR INABILITY TO USE THE PRODUCT, (ii) THE COST OF ANY SUBSTITUTE PRODUCTS AND/OR SERVICES RESULTING FROM ANY PRODUCTS, DATA, INFORMATION OR SERVICES OBTAINED OR WHICH YOU WERE UNABLE TO OBTAIN OR TRANSACTIONS EFFECTED OR FAILED TO BE EFFECTED, (iii) THE USE OR INABILITY TO USE ANY THIRD PARTY APPLICATIONS CONTAINED WITHIN THE PRODUCT, OR (iv) ANY MATTER OTHERWISE RELATED TO YOUR USE OF THE PRODUCT.
Release of Liability
You assume all risks associated with use of the Product including, but not limited to any harm, injury or damages resulting directly or indirectly from the use of the Product, all such risks being known and understood by You. In consideration of your use of the Product, You, for yourself and anyone entitled to act on your behalf, waive and forever release ACCF, its officers, trustees, employees, representatives and successors from all claims and liabilities of any kind arising out of your use or misuse of the Product.
Indemnification
You hereby agree to indemnify, save and hold ACCF, its directors, officers, shareholders, parents, subsidiaries, affiliates, agents and licensors harmless from and against any and all claims, liability, losses, damages and costs, including, without limitation, reasonable attorneys' fees and costs, arising out of your use or misuse of the Product or Content, or any violation of this Agreement. ACCF assumes the right, at your expense, to assume the exclusive defense and control of any matter for which you are required to indemnify ACCF, and you agree to cooperate with ACCF’s defense of these claims. ACCF will use reasonable efforts to notify you of any such claim, action, or proceeding upon becoming aware of it.
Disclaimer of Warranties
THE PRODUCT AND CONTENT ARE PROVIDED ON AN "AS IS" AND "AS AVAILABLE" BASIS. ACCF AND ITS SUPPLIERS EXPRESSLY DISCLAIM ALL WARRANTIES OF ANY KIND WITH RESPECT TO THE PRODUCT OR CONTENT, WHETHER EXPRESS OR IMPLIED, INCLUDING IMPLIED WARRANTIES OF MERCHANTABILITY, FITNESS FOR A PARTICULAR PURPOSE, TITLE AND NON-INFRINGEMENT. ACCF MAKES NO WARRANTY THAT THE PRODUCT AND/OR ANY CONTENT THEREIN WILL MEET YOUR REQUIREMENTS, OR WILL BE UNINTERRUPTED, TIMELY, SECURE, CURRENT, ACCURATE, COMPLETE OR ERROR-FREE OR THE RESULTS THAT MAY BE OBTAINED BY USE OF THE PRODUCT OR ANY CONTENT THEREIN WILL BE ACCURATE OR RELIABLE. YOU UNDERSTAND AND ACKNOWLEDGE THAT YOUR SOLE AND EXCLUSIVE REMEDY WITH RESPECT TO ANY DEFECT IN OR DISSATISFACTION WITH THE PRODUCT IS TO CEASE ITS USE.
The Content on the Product is presented as an educational service intended for licensed healthcare professionals. While the Content in the Product is about specific medical and healthcare issues, the Content is not a substitute for or replacement of personalized medical advice and is not intended to be used as the sole basis for making individualized medical or health-related decisions.
The views and opinions expressed are those of the contributing authors and editors and do not necessarily represent the views of the ACCF. The material is not intended to present the only, or necessarily best, methods or procedures for the medical situations addressed, but rather is intended to represent an approach, view, statement or opinion.
Any reference to a specific therapy or commercial product in this Product does not constitute a guarantee or endorsement by ACCF of the quality or value of such therapy or product or any claims made by the manufacturer of such therapy or commercial product.
In addition, any statements about such therapy or commercial products are solely based on published clinical prediction rules and estimates of drug treatment effects from published clinical studies and do not represent an ACCF endorsement or evaluation of these products.
Force Majeure
ACCF will be excused from performance under this Agreement and will not be liable or considered in default under this Agreement in the event that the Product is unavailable for any period of time, or if ACCF is otherwise unable to perform its obligations hereunder, in whole or in part, as a result of a Force Majeure Event. For purposes of this Section, "Force Majeure Event" means an event or series of events caused by or resulting from any of the following: (1) weather conditions or other elements of nature or acts of God; (2) government regulation; (3) quarantines or embargoes; (4) telecommunications, network, computer, server or Internet downtime; (5) unauthorized access to ACCF's information technology systems by third parties; or (6) any other causes beyond the reasonable control of ACCF.
No Assignment
This Agreement is personal to You, and You may not assign your rights or obligations to anyone.
No Waiver
Neither failure nor delay on the part of any party to exercise any right, remedy, power or privilege hereunder nor course of dealing between the parties shall operate as a waiver thereof, or of the exercise of any other right, remedy, power or privilege. No term of this Agreement shall be deemed waived, and no breach consented to, unless such waiver or consent shall be in writing and signed by the party claimed to have waived or consented. No waiver of any rights or consent to any breaches shall constitute a waiver of any other rights or consent to any other breach.
Severability
If any provision in this Agreement is held invalid or unenforceable under applicable law, the remaining provisions shall continue in full force and effect.
Governing Law
This Agreement will be governed by and construed exclusively in accordance with the laws of the District of Columbia, USA, without regard to its conflicts of law principles and, to the extent applicable, the federal laws of the United States. If a dispute arises between ACCF and You, You hereby agree to submit such dispute to non-binding mediation, followed by binding arbitration, if necessary. Both the mediation and arbitration will be conducted by JAMS applying the laws of the District of Columbia without regard to its conflicts of laws principles and in the District of Columbia as venue.
Certification
I hereby certify that I understand and agree to the terms stated in this Agreement and that this Agreement applies to my initial use of the Product and all other subsequent uses of the Product.
BY USING THIS PRODUCT, I HEREBY AFFIRM THAT I HAVE READ, FULLY UNDERSTAND, AND AGREE TO THE ABOVE STATEMENTS.
About the App
When was this App last updated?
January 2024
How can I provide feedback?
How is the ManageMR app intended to be used?
ManageMR provides clinicians with personalized referral and treatment advice for patients diagnosed with mitral regurgitation by verifying the severity and etiology of the MR. For optimal use:
- Conduct a clinical assessment of your patient with suspected chronic mitral regurgitation, including assessment of qualitative and quantitative parameters, symptoms, exercise tolerance, physical exam and chamber sizes.
-
- Use the MR Echo Reporting Checklist tool to help review and report echo parameters in a standardized way. A link to this tool can be found in the orange introductory box of the app, or here
- Using the results of the initial assessment, input the required patient parameters into the Evaluate screen of the app.
- Review app’s advice on the Advice screen regarding:
-
- - suspected level and probability of MR severity
- - indicated mechanism and etiology of MR
- - whether additional testing is suggested
- Consider the advice in conjunction with initial clinical assessment.
- If patient’s MR is severe, review suggestions on the Advice screen regarding intervention next steps for your patient, and use it to help inform treatment decisions.
- Based on the definition of your patient’s MR, use the app’s Advice screen to help determine if monitoring or referral to additional experts is suggested for your patient. If need for referral is determined by the clinician, they can use the MR Referral Checklist tool to help facilitate a comprehensive referral handoff.
- Use the Email button on the app’s Advice tab to email yourself a summary of the advice given by the app.
- Visit the Resources tab in the app for
-
- - additional reference material regarding the management of MR
- - more clinician tools to help implement guidance from the ACC/AHA Valvular Heart Disease Guideline and ACC’s Mitral Regurgitation Expert Consensus Clinical Pathway
- - patient tools to help patient’s monitor and manage their own MR
The information and recommendations in this App are meant to support clinical decision making. They are not meant to represent the only or best course of care, or replace clinical judgment. Therapeutic options should be determined after discussion between the patient and their care provider.
How was the ManageMR app developed?
The content for this App is derived from the 2020 ACC Focused Update to the 2017 ACC Expert Consensus Decision Pathway on the Management of Mitral Regurgitation. App content and design was further refined and vetted by ACC member clinicians, and through user testing with clinicians practicing in relevant specialties.
This app was developed as part of a continuing initiative to enable clinicians to access and implement ACC clinical policy at the point of care for better patient care and outcomes. Previous versions of the app were developed as part of the ACC's Emerging Mitral Regurgitation Clinical Care (EMC2) Initiative, and was supported by Founding Sponsor Abbott Vascular. All content was independently developed with no sponsor involvement.
Where can I go if I have questions?
For Support
Call: (202) 375-6000, ext. 5603 or (800) 253-4636
Email: MemberCare@acc.org
Locked
News