- Recent ACS (within the past 12 mo.)
- History of MI (other than recent ACS event listed above)
- History of ischemic stroke
- Symptomatic PAD (history of claudication with ABI <0.85, or previous revascularization or amputation)
Where is the ASCVD Risk Estimator?
Clinician Resources - General
Clinician Resources - LDL-C Lowering Therapy
Clinician Resources - Hypertriglyceridemia
Clinician Resources - Statin Intolerance
Other mobile tools
References
-
Blumenthal, R, Morris, P, Gaudino, M. et al.
2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia:
A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice
Guidelines. JACC. 2026.
https://doi.org/10.1016/j.jacc.2025.11.016 -
Eckel, R, Jakicic, J, Ard, J. et al.
2013 AHA/ACC Guideline on Lifestyle Management to Reduce Cardiovascular Risk:
A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. JACC. 2014 Jul, 63 (25_Part_B) 2960–2984.
https://doi.org/10.1016/j.jacc.2013.11.003 -
Jensen, M, Ryan, D, Apovian, C. et al.
2013 AHA/ACC/TOS Guideline for the Management of Overweight and Obesity in Adults:
A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The
Obesity Society. JACC. 2014 Jul, 63 (25_Part_B) 2985–3023.
https://doi.org/10.1016/j.jacc.2013.11.004 -
Khan SS, Matsushita K, Sang Y, et al.
Development and Validation of the American Heart Association Predicting Risk of Cardiovascular Disease EVENTs (PREVENT™) Equations
Circulation 2023.
DOI: 10.1161/CIRCULATIONAHA.123.067626.
-
Khan SS, Coresh J, Pencina MJ, et al.
Novel Prediction Equations for Absolute Risk Assessment of Total Cardiovascular Disease Incorporating
Cardiovascular-Kidney-Metabolic Health:
A Scientific Statement From the American Heart Association. Circulation 2023;148(24):1982-2004.
DOI: 10.1161/CIR.0000000000001191.
- Social deprivation index (SDI) Robert Graham Center - Policy Studies in Family Medicine & Primary Care. (2018, November 5). Retrieved November 29, 2021, from https://www.graham-center.org/rgc/maps-data-tools/social-deprivation-index.html
Major ASCVD Events
High-risk Conditions
- Age ≥ 65 y
- Heterozygous familial hypercholesterolemia
- History of prior coronary artery bypass surgery or percutaneous coronary intervention outside of the major ASCVD event(s)
- Diabetes
- Hypertension
- CKD (eGFR 15-59 mL/min/1.73 m²)
- Current smoking
- Persistently elevated LDL-C (LDL-C ≥100 mg/dL [≥2.6 mmol/L]) despite maximally tolerated statin therapy and ezetimibe
- History of congestive HF
*Very high-risk includes a history of multiple major ASCVD events or 1 major ASCVD event and multiple high-risk conditions.
- ABI = ankle-brachial index
- ACS = acute coronary syndrome
- ASCVD = atherosclerotic cardiovascular disease
- CKD = chronic kidney disease
- eGFR = estimated glomerular filtration rate
- HF = heart failure
- LDL-C = low-density lipoprotein cholesterol
- MI = myocardial infarction
- PAD = peripheral artery disease
Risk-Enhancing Factors
- Family history of premature ASCVD (males, age <55 y; females age <65 y)
- Primary hypercholesterolemia (LDL-C 160–189 mg/dL [4.1–4.8 mmol/L); non–HDL-C 190–219 mg/dL [4.9–5.6 mmol/L] *
- Metabolic syndrome (increased waist circumference, elevated triglycerides [≥150 mg/dL], elevated blood pressure, elevated glucose, and low HDL-C [ <40 mg/dL in men; <50 mg/dL in women] are factors; tally of 3 makes the diagnosis
- Chronic kidney disease (eGFR 15–59 mL/min/1.73 m² with or without albuminuria; not treated with dialysis or kidney transplantation)
- Chronic inflammatory conditions such as psoriasis, RA, or HIV/AIDS
- History of premature menopause (before age 40 y) and history of pregnancy-associated conditions that increase later ASCVD risk such as preeclampsia
- High-risk race/ethnicities (e.g., South Asian ancestry)
- Lipid/biomarkers: Associated with increased ASCVD risk
o Persistently* elevated, primary hypertriglyceridemia (≥175 mg/dL)
o If measured:- 1. Elevated high-sensitivity C-reactive protein (≥2.0 mg/L)
2. Elevated Lp(a): A relative indication for its measurement is family history of premature ASCVD. An Lp(a) ≥50 mg/dL or ≥125 nmol/L constitutes a risk-enhancing factor especially at higher levels of Lp(a).
3. Elevated apoB ≥130 mg/dL: A relative indication for its measurement would be triglyceride ≥200 mg/dL. A level ≥130 mg/dL corresponds to an LDL-C≥160 mg/dL and constitutes a risk-enhancing factor
4. ABI <0.9
* Optimally, 3 determinations
Abbreviations:
- AIDS = acquired immunodeficiency syndrome
- ABI = ankle-brachial index
- apoB = apolipoprotein B
- ASCVD = atherosclerotic cardiovascular disease
- eGFR = estimated glomerular filtration rate
- HDL-C = high-density lipoprotein cholesterol
- HIV = human immunodeficiency virus
- LDL-C = low-density lipoprotein cholesterol
- Lp(a) = lipoprotein (a)
- RA = rheumatoid arthritis
Factors to Consider in the Clinician–Patient Discussion
Potential for additional ASCVD risk reduction from addition of nonstatin therapy to evidence-based statin therapy to lower LDL-C
- Percentage LDL-C reduction achieved with evidence-based statin therapy (if < 50% and not on maximally tolerated statin, should increase statin therapy first and reinforce lifestyle modifications) and whether patient is above LDL-C threshold for consideration of nonstatin therapies
- For patients with ASCVD, patient’s status as very high risk or not very high risk on evidence-based statin therapy (See Criteria for Defining Patients at Very High-Risk of future ASCVD Events)*
- For patients without ASCVD or baseline LDL-C ≥190 mg/dL, patient’s baseline predicted 10-year ASCVD risk pre-statin and presence of risk enhancing factors (See Risk-Enhancing Factors for Clinician–Patient Risk Discussion)†
- Available scientific evidence of ASCVD risk reduction (and magnitude of benefit) when nonstatin therapy is added to evidence-based statin therapy‡
- Additional desired % LDL-C lowering beyond that achieved on evidence-based statin therapy§
- Mean percentage LDL-C lowering expected with proposed nonstatin therapy when added to evidence-based statin therapy
Potential for clinically significant adverse events or drug-drug interactions from addition of nonstatin therapy to evidence-based statin therapy for lowering LDL-C
- See Strategies and Nonstatin Agents Considered for Management of LDL-Related ASCVD Risk.
Cost considerations
- Potential out-of-pocket cost of therapy to the patient (e.g., insurance plan coverage, pharmacy or medical benefit, copayment, availability of assistance programs).
Patient preferences and considerations
- Patient’s perception of benefit from addition of nonstatin therapy.
- Convenience of nonstatin therapy (e.g., route, setting [home or medical office], and frequency of administration, pill burden, storage).
- Potential of nonstatin therapy to jeopardize adherence to other evidence-based therapies.
- Cost of nonstatin therapy.
- Anticipated life expectancy, comorbidities, and impact of therapy on quality of life.
∗ For example, in the Treating to New Targets trial, patients with CHD who received 10 mg of atorvastatin daily had a 5-y event rate of 10.9%, and those who received 80 mg of atorvastatin daily had a 5-y event rate of 8.7%. These numbers (and similar rates from other trials) may inform the number-needed-to-treat. Additional consideration of comorbidities and other poorly controlled or well-controlled risk factors will increase or decrease risk accordingly. See Table 1 for criteria for defining patients at very high risk.
† Use the Pooled Cohort Equations to estimate 10-y ASCVD risk. See Table 1 for criteria for defining patients at very high risk.
‡ Such evidence exists for ezetimibe from the IMPROVE-IT study, with a 6% relative/2% absolute risk reduction in a composite ASCVD endpoint over 7 y when added to a moderate-intensity statin. Evidence from FOURIER and ODYSSEY Outcomes demonstrate 2% absolute/15% relative ASCVD risk reduction. Data are lacking for addition of BAS to statins, bempedoic acid, inclisiran, and evinacumab. Niacin preparations have been associated with no benefit and potential for significant harms when added to statin therapy.
§ For example, patients on maximally-tolerated statin therapy with LDL-C ≥130 mg/dL may receive more benefit from the addition of a nonstatin therapy than those with on-statin LDL-C of 80 mg/dL.
‖For example, when added to statins, ezetimibe may lower LDL-C an additional 20%-25% on average; PCSK9 inhibitors may lower LDL-C an additional 60% on average. For each 40-mg/dL reduction in LDL-C using safe and evidence-based therapies, there appears to be an approximate 20% relative risk reduction in ASCVD. This number, combined with the baseline absolute risk, may inform the number-needed-to-treat.
Abbreviations:- ASCVD = atherosclerotic cardiovascular disease
- LDL-C = low-density lipoprotein cholesterol
Referral to another clinician
Referral to lipid specialist
- Consider referring any patient with ASCVD and/or baseline LDL-C ≥190 mg/dL, baseline LDL-C ≥190 mg/dL, or intolerance to at least 2 (preferably 3) statin therapies with 1 attempt at the lowest FDA-approved dose and a trial of an alternative statin therapy regimen (eg, every-other-day dosing)
- Referral is recommended for patients with ASCVD and baseline LDL-C ≥190 mg/dL who did not achieve ↓ LDL-C ≥50% and LDL-C <70 mg/dL (or non–HDL-C <100 mg/dL) on maximally tolerated statin therapy in combination with nonstatin therapy
- May also consider referring other patients unable to achieve adequate LDL-C reduction
- Considerations in referring: Lipid specialists may be available for virtual visits for patients in some rural or remote locations
Referral to RD/RDN
- Consider referring any patient with ASCVD and/or baseline LDL-C ≥190 mg/dL, or baseline LDL-C ≥190 mg/dL
- Referral is recommended for patients with ASCVD and baseline LDL-C ≥190 mg/dL who did not achieve ↓ LDL-C ≥50% and LDL-C <70 mg/dL (or non–HDL-C <100 mg/dL) on maximally tolerated statin therapy in combination with nonstatin therapy
- May also consider referring other patients unable to achieve adequate LDL-C reduction
Non-statin agents that may be used to manage LDL-related ASCVD risk
Ezetimibe
- Mechanism of action: Inhibits NPC1L1 protein; reduces cholesterol absorption in small intestine.
-
FDA-approved indication(s): As adjunct to diet to:
- ↓ TC, LDL-C, ApoB, non–HDL-C in patients with primary hyperlipidemia, either alone or in combination with statin therapy;
- ↓ TC, LDL-C, ApoB, non–HDL-C in patients with mixed hyperlipidemia in combination with fenofibrate;
- ↓ TC, LDL-C with HoFH, in combination with atorvastatin or simvastatin;
- ↓ sitosterol and campesterol in patients with homozygous sitosterolemia (phytosterolemia)
- Dose: 10 mg orally daily, with or without food. Take either ≥2 h before or ≥4 h after BAS, if used in combination
- Mean % reduction in LDL-C (per PI): Monotherapy—18%; combination therapy with statin therapy (incremental reduction)—25%
- Contraindication: History of hypersensitivity to this medication.
-
Warnings/precautions:
- Not recommended in patients with moderate/severe hepatic impairment.
- Persistent elevations in hepatic transaminases may occur with concomitant statin therapy. Monitor hepatic transaminases before and during treatment based on monitoring recommendations for statin therapy.
- Cases of myopathy and rhabdomyolysis have been reported when ezetimibe was used alone or in combination with statin therapy.
- Adverse effects: Monotherapy—upper respiratory tract infection, diarrhea, arthralgia, sinusitis, pain in extremities. In combination with statin—nasopharyngitis, myalgia, upper respiratory tract infection, arthralgia, diarrhea
- Use during pregnancy/lactation: No safety data in humans; avoid use
- Drug–drug interactions: Cyclosporine, fibrates, BAS
- CV outcomes trials: IMPROVE-IT (The addition of ezetimibe to moderate-intensity statin therapy in patients with recent ACS resulted in incremental lowering of LDL-C and reduced the primary composite endpoint of CV death, nonfatal MI, UA requiring rehospitalization, coronary revascularization [≥30 days after randomization], or nonfatal stroke. The median follow-up was 6 years); SHARP (Simvastatin plus ezetimibe reduced LDL-C and reduced the primary endpoint of first major ASCVD event [nonfatal MI or CHD death, nonhemorrhagic stroke, or any arterial revascularization procedure] compared with placebo in patients with CKD over a median follow-up of 4.9 years)
- Other prescribing considerations: Generally well tolerated. Generic available
PCSK9 mAb (Alirocumab, Evolocumab)
- Mechanism of action: Human mAb to PCSK9. Binds to PCSK9 and increases the number of LDL receptors available to clear circulating LDL-C
- FDA-approved indication(s):
- Alirocumab and evolocumab:
- ↓ LDL-C in adults with primary hyperlipidemia (including HeFH) as adjunct to diet, either alone or in combination with other lipid-lowering therapies
-
Alirocumab:
- ↓ risk of MI, stroke, and unstable angina requiring hospitalization in adults with ASCVD;
- ↓ LDL-C in adults with HoFH as adjunct to other LDL-C–lowering therapies
-
Evolocumab:
- ↓ risk of MI, stroke, and coronary revascularization in adults with ASCVD;
- ↓ LDL-C in pediatric patients (aged ≥10 years) with HeFH as adjunct to diet and other LDL-C–lowering therapies;
- ↓ LDL-C in adults and pediatric patients (aged ≥10 years) with HoFH as adjunct to diet and other LDL-C–lowering therapies
- Dose and route of administration:
- Alirocumab: Administer SC in the thigh, abdomen, or upper arm. In adults with ASCVD or primary hyperlipidemia: initiate 75 mg SC every 2 weeks. If more LDL-C reduction needed, may ↑ dose to 150 mg every 2 weeks. Alternative starting dose is 300 mg SC every 4 weeks. For the 300-mg dose, administer 2 (150-mg) injections consecutively at 2 different injection sites. In adults with HeFH undergoing LDL apheresis or adults with HoFH, administer 150 mg SC every 2 weeks
- Evolocumab: Administer SC in the thigh, abdomen, or upper arm. In adults with ASCVD, adults with primary hypercholesterolemia, including with established clinical ASCVD or HeFH, or in pediatric patients (aged ≥10 years) with HeFH, administer 140 mg SC every 2 weeks or 420 mg SC once monthly in abdomen, thigh, or upper arm. In adults or pediatric patients (aged ≥10 years) with HoFH, administer 420 mg SC once monthly; if more LDL-C reduction is needed after 12 weeks, may ↑ dose to 420 mg every 2 weeks. In adults or pediatric patients (age ≥10 years) with HoFH on LDL apheresis, may initiate 420 mg SC every 2 weeks to correspond with apheresis schedule; evolocumab should be given after apheresis is complete. To administer 420-mg dose, either use the prefilled single-dose on-body infuser or give 3 (140-mg) injections consecutively within 30 min.
- Mean % LDL-C reduction (per PI):
- Alirocumab: when added to maximally tolerated statin therapy, alirocumab 75 mg and 150 mg SC every 2 weeks ↓ LDL-C by an additional 45% and 58%, respectively, when added to maximally tolerated statin therapy.
- Evolocumab: 140 mg every 2 weeks and 420 mg SC every 4 weeks, ↓ LDL-C by an additional 64% and 58%, respectively.
- Contraindication: History of hypersensitivity to the medication.
- Warnings/precautions: Hypersensitivity reactions occurred during clinical trials. If a serious hypersensitivity reaction occurs, discontinue therapy; treat according to standard of care; monitor until signs and symptoms resolve.
- Adverse effects:
- Alirocumab: In patients with primary hyperlipidemia: nasopharyngitis, injection site reactions, influenza; in patients with ASCVD: noncardiac chest pain, nasopharyngitis, myalgia. No evidence of increase in cognitive adverse effects observed in ODYSSEY Outcomes or CANTAB.
- Evolocumab: In patients with primary hyperlipidemia: nasopharyngitis, upper respiratory tract infection, influenza, back pain, and injection site reactions; in patients with ASCVD: diabetes, nasopharyngitis, upper respiratory tract infection.
- No evidence of an increase in cognitive adverse effects observed in FOURIER or EBBINGHAUS.
- Use during pregnancy/lactation: No safety data in humans; avoid use.
- Drug-drug interactions: No clinically significant drug-drug interactions identified for alirocumab or evolocumab
- CV outcomes trials:
- Alirocumab: ODYSSEY Outcomes in 18,600 post-ACS (4-52 weeks) patients on evidence-based statin therapy; Demonstrated that addition of alirocumab reduced the primary endpoint of CHD death, MI, ischemic stroke, or hospitalization for UA.
- Evolocumab: FOURIER in 27,564 patients with prior MI, stroke, or PAD on atorvastatin ≥20 mg or equivalent; Demonstrated that addition of evolocumab reduced the primary endpoint of CV death, MI, stroke, revascularization, or hospitalization for unstable angina.
- Other prescribing considerations: Robust LDL-C reduction, cost, SC administration at home, may require prior authorization.
- Evolocumab: Advise latex-sensitive patients that the needle covers on the products contain latex.
Bempedoic acid
- Mechanism of action: ACL inhibitor; inhibits cholesterol synthesis in the liver; increases LDL receptor density. Bempedoic acid and its active metabolite require coenzyme A activation by ACSVL1, which is expressed primarily in the liver.
- FDA-approved indication(s): ↓ LDL-C in adults with ASCVD or HeFH as adjunct to diet and maximally tolerated statin therapy.
- Dose: 180 mg orally once daily, with or without food.
- Mean % reduction in LDL-C (per PI): Combination therapy with statin therapy (placebo-corrected incremental reduction)—17%-18%.
- Contraindication: none
-
Warnings/precautions:
- May ↑ serum uric acid. Advise patients to contact their clinician if symptoms of hyperuricemia occur. Assess serum uric acid when clinically indicated. Monitor patients for signs and symptoms of hyperuricemia, and initiate treatment with urate-lowering drugs, as appropriate. Assess uric acid level before initiation and if signs and symptoms of hyperuricemia occur.
- Discontinue immediately if the patient experiences rupture of a tendon. Consider discontinuing if the patient experiences joint pain, swelling, or inflammation. Advise patients to rest at the first sign of tendinitis or tendon rupture and to contact their health care provider if tendinitis or tendon rupture symptoms occur. Consider alternative therapy in patients with a history of tendon disorders or tendon rupture.
- Adverse effects: Upper respiratory tract infection, muscle spasms, hyperuricemia, back pain, abdominal pain or discomfort, bronchitis, pain in extremity, anemia, elevated liver enzymes.
- Use during pregnancy/lactation: Discontinue when pregnancy is recognized unless the benefits of therapy outweigh the potential risks to the fetus. There are no available data on use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
- Drug–drug interactions: Avoid concomitant simvastatin >20 mg daily or pravastatin >40 mg daily.
- CV outcomes trials: CV outcomes trials not completed. CLEAR Outcomes trial completion expected later in 2022.
- Other prescribing considerations: cost; pill burden; requires prior authorization
Bempedoic acid and ezetimibe
- Refer to section on ezetimibe for information specific to this agent.
- Mechanism of action: See the mechanisms of action for bempedoic acid and ezetimibe included in this table.
- FDA-approved indication(s): ↓ LDL-C in adults with ASCVD or HeFH as adjunct to diet and maximally tolerated statin therapy.
- Dose: 1 tablet (180 mg bempedoic acid/10 mg ezetimibe) orally, once daily, with or without food. Swallow whole. Take either ≥2 hours before or ≥4 hours after BAS, if used in combination.
- Mean % reduction in LDL-C (per PI): Combination therapy with statin therapy (placebo-corrected incremental reduction)—38%.
- Contraindication: History of hypersensitivity to ezetimibe.
-
Warnings/precautions:
- May ↑ serum uric acid. Advise patients to contact their clinician if symptoms of hyperuricemia occur. Assess serum uric acid when clinically indicated. Monitor patients for signs and symptoms of hyperuricemia, and initiate treatment with urate-lowering drugs as appropriate. Assess uric acid level before initiation and if signs and symptoms of hyperuricemia occur.
- Discontinue immediately if the patient experiences tendon rupture. Consider discontinuing if the patient experiences joint pain, swelling, or inflammation. Advise patients to rest at the first sign of tendinitis or tendon rupture and to contact their health care provider if tendinitis or tendon rupture symptoms occur. Consider alternative therapy in patients with a history of tendon disorders or tendon rupture.
- Adverse effects: Upper respiratory tract infection, muscle spasms, hyperuricemia, back pain, abdominal pain or discomfort, bronchitis, pain in extremities, anemia, elevated liver enzymes, diarrhea, arthralgia, sinusitis, fatigue, influenza. Consider alternative therapy if history of tendon disorder or rupture; discontinue immediately if tendon rupture occurs.
- Use during pregnancy/lactation: Discontinue when pregnancy is recognized unless the benefits of therapy outweigh the potential risks to the fetus. There are no available data on use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
- Drug–drug interactions: Cyclosporine; fibrates. Avoid concomitant simvastatin >20 mg daily or pravastatin >40 mg daily.
- CV outcomes trials: CV outcomes trials for bempedoic acid not completed. Completion of CLEAR Outcomes trial expected later in 2022. CV outcomes trial will not be required for fixed-dose combination of ezetimibe and bempedoic acid.
- Prescribing considerations: ↓ LDL-C within the range of moderate-intensity statin therapy; cost; requires prior authorization
Inclisiran
- Mechanism of action: siRNA targeting PCSK9; inhibits PCSK9 production in liver, thereby prolonging activity of LDL receptors.
- FDA-approved indication(s): ↓ LDL-C in adults with ASCVD or HeFH as adjunct to diet and maximally tolerated statin therapy.
- Dose: Administer 284 mg SC on day 1, day 90, and then every 6 months by a clinician.
- Mean % reduction in LDL-C (per PI): 48%-52%
- Contraindications (per PI): None
- Warnings/precautions (per PI): None
- Adverse effects: Injection site reaction, arthralgia, urinary tract infection, diarrhea, bronchitis, pain in extremities, dyspnea
- Use during pregnancy/lactation: No safety data in humans; avoid use.
- Drug–drug interactions (per PI): None
- CV outcomes trials: CV outcomes trials not yet completed. ORION-4 currently in progress with estimated completion in 2026. VICTORION-2P currently in progress with estimated completion in 2027.
- Other prescribing considerations: robust LDL-C reduction, cost, requires SC administration by a clinician, requires prior authorization.
BAS
- Mechanism of action: Nonabsorbed, lipid-lowering polymer that binds bile acids in the intestine and impedes their reabsorption. As the bile acid pool ↓, the hepatic enzyme cholesterol 7-a-hydroxylase is up-regulated, which ↑ conversion of cholesterol to bile acids. This causes ↑ demand for cholesterol in the liver cells, resulting in the dual effect of increasing transcription and activity of the cholesterol biosynthetic enzyme HMG-CoA reductase and ↑ numbers of hepatic LDL receptors. These compensatory effects result in ↑ clearance of LDL particles from the blood, in turn resulting in ↓ serum LDL-C levels. Serum TG levels may ↑ or remain unchanged.
- FDA-approved indication(s):
- Colesevelam: As an adjunct to diet and exercise
- ↓ LDL-C in adults with primary hyperlipidemia;
- ↑ glycemic control in adults with type 2 diabetes;
- ↓ LDL-C in boys and post-menarchal girls (aged 10-17 years) with HeFH who are unable to reach LDL-C targets after an adequate trial of diet therapy and lifestyle modifications. Cholestyramine, colestipol: ↓ LDL-C with primary hyperlipidemia, as adjunct to diet
- Dose and route of administration:
-
Colesevelam: Tablets: 6 tablets orally once daily or 3 tablets orally twice
daily; take tablets with a meal and liquid.
Suspension: one 3.75-g packet orally daily, or one 1.875-g packet orally twice daily; mix powder with 8 ounces of water, fruit juice, or soft drink; take with meal. - 3.75 g is equivalent to 6 tablets. 1.875 g is equivalent to 3 tablets;
- Cholestyramine: 8-16 g/day orally, divided into 2 doses;
- Colestipol: 2-16 g/day orally, given once or in divided doses
- Mean % LDL reduction (per PI):
- Colesevelam: Monotherapy—15% (6 tablets daily); in combination with low- to moderate-intensity statin therapy—additional 10%-16% reduction in LDL-C (data from simvastatin 10 mg, atorvastatin 10 mg). Cholestyramine: Monotherapy—10.4% vs placebo.
- Colestipol: not provided in PI. In dose-ranging RCT with monotherapy, doses of 5, 10, and 15 g resulted in 16.3%, 22.8%, and 27.2% reductions in LDL-C, respectively
- Contraindications (per PI):
- Colesevelam: TG >500 mg/dL; history of hypertriglyceridemia-induced pancreatitis; bowel obstruction.
- Cholestyramine: History of serious hypersensitivity to this medication.
- Use during pregnancy/lactation: Considered safe to use
- Colestipol: Complete biliary obstruction, history of serious hypersensitivity to this medication.
- Warnings/precautions: May ↑ TG and cause acute pancreatitis, monitor TG, discontinue if signs and symptoms of acute pancreatitis occur; may cause GI obstruction, avoid with gastroparesis, other GI motility disorders, and history of major GI tract surgery with risk for bowel obstruction; may cause vitamin K or fat-soluble vitamin deficiencies, oral vitamins should be given ≥4 hours before this medication; may decrease absorption of other medications, other medications should be given ≥4 hours before this medication. Some products contain phenylalanine, which may be harmful to patients with phenylketonuria.
- Adverse effects: Constipation, dyspepsia, and nausea.
- Use during pregnancy/lactation: Considered safe to use
- Drug-drug interactions: In general, BAS may decrease absorption of other medications; it is a good practice for all other medications to be given ≥4 hours before BAS. Concomitant use of BAS is known to decrease absorption of cyclosporin, oral contraceptives containing ethinyl estradiol and norethindrone, olmesartan, phenytoin, sulfonylureas, thyroid replacement therapy, warfarin; give these medications ≥4 hours before BAS. For patients on warfarin, monitor INR frequently during BAS initiation and then periodically. Cholestyramine may increase exposure to metformin; monitor glycemic control.
- CV outcomes trials: In LRC-CPPT, 3,806 asymptomatic middle-aged men with primary hypercholesterolemia were randomized to cholestyramine resin vs placebo for an average of 7.4 years. The cholestyramine group experienced a 19% reduction in risk (P < 0.05) of the primary endpoint—definite CHD death and/or definite nonfatal MI. The effects of colesevelam and colestipol on cardiovascular morbidity and mortality have not been determined
- Considerations in prescribing: Pill burden; inconvenience in preparation of oral suspension preparations; drug interactions, GI side effects; exacerbation of hypertriglyceridemia; orally administered, colesevelam lowers HbA1c 0.5% in diabetes; CV outcomes data not available for all products
Agents that may be used to treat HoFH under care of a lipid specialist
Evinacumab
- Mechanism of action: Human monoclonal antibody that binds to and inhibits ANGPTL3. Promotes VLDL processing and clearance upstream of LDL formation
- FDA-approved indication(s): ↓ LDL-C in adults and pediatric patients (aged ≥12 years) with HoFH as adjunct to other LDL-C–lowering therapies
- Dose and route of administration: 15 mg/kg administered by healthcare professional as IV infusion once monthly (every 4 weeks). See PI for preparation and administration instructions.
- Mean % reduction in LDL-C (per PI): Combination therapy with other lipid-lowering therapies (incremental reduction)—49%.
- Contraindication: History of serious hypersensitivity to this medication.
-
Warnings/precautions:
- Hypersensitivity reactions occurred during clinical trials. If a serious hypersensitivity reaction occurs, discontinue therapy; treat according to standard of care; monitor until signs and symptoms resolve.
- May cause fetal toxicity; inform patients who may become pregnant of risk to fetus; obtain a pregnancy test before initiating therapy in patients who may become pregnant; advise patients who may become pregnant to use contraception during treatment and for ≥5 months following the last dose. Discontinue this medication if patient becomes pregnant. Clinicians should report pregnancies that occur while taking this medication (1-833-385-3392).
- Adverse effects: nasopharyngitis, influenza-like illness, dizziness, rhinorrhea, nausea.
- Use during pregnancy/lactation: Avoid use.
- Drug–drug interactions: No clinically significant drug-drug interactions have been identified
- CV outcomes trials: The effect of evinacumab on CV morbidity and mortality has not been determined
- Other prescribing considerations: See prescribing information for complete preparation and administration instructions. Robust LDL-C reduction; cost, IV administration, requires prior authorization
Lomitapide
- Mechanism of action: Directly binds and inhibits microsomal triglyceride transfer protein, which resides in the lumen of the endoplasmic reticulum, thereby preventing the assembly of apoB-containing lipoproteins in enterocytes and hepatocytes. This inhibits synthesis of chylomicrons and VLDL and leads to ↓ LDL-C
- FDA-approved indications: ↓ LDL-C, TC, apoB, and non–HDL-C in patients with HoFH, as adjunct to a low-fat diet and other lipid-lowering treatments (including LDL apheresis, where available)
- Dose and route of administration: Initiate 5 mg orally once daily. Titrate dose based on acceptable safety/tolerability: increase to 10 mg daily after at least 2 weeks and then, at a minimum of 4-week intervals, to 20 mg, 40 mg, up to the maximum recommended dose of 60 mg daily
- Mean % LDL reduction (per PI): Mean and median percent changes in LDL-C from baseline when added to baseline lipid-lowering therapy were -40% and -50%, respectively
-
Black box warnings:
- May cause elevations in liver transaminases; measure ALT, AST, alkaline phosphatase, total bilirubin before initiating this medication; during treatment, adjust dose if ALT or AST ≥3 times the upper limit of normal; discontinue this medication for clinically significant liver toxicity.
- Increases hepatic fat (hepatic steatosis) with or without concomitant increases in transaminases. Hepatic steatosis associated with lomitapide may be a risk factor for progressive liver disease, including steatohepatitis and cirrhosis. Because of the risk of hepatotoxicity, lomitapide is only available through the REMS program
-
Contraindications:
- Pregnancy.
- concomitant use with strong/moderate CYP3A4 inhibitors;
- moderate/severe hepatic impairment or active liver disease including unexplained persistent abnormal liver function tests.
-
Warnings/precautions:
- May cause fetal toxicity; inform patients who may become pregnant of risk to fetus; obtain a pregnancy test before initiating therapy in patients who may become pregnant; advise patients who may become pregnant to use contraception during treatment and for ≥2 weeks following the last dose. Discontinue this medication if patient becomes pregnant. Clinicians should report pregnancies that occur while taking this medication (1-877-902-4099).
- Adverse effects: Diarrhea, nausea, vomiting, dyspepsia, and abdominal pain.
- Use during pregnancy/lactation: Avoid use.
-
Drug–drug interactions:
- CYP3A4 inhibitors increase exposure to lomitapide. Strong/moderate CYP3A4 inhibitors are contraindicated with lomitapide. Avoid grapefruit juice.
- Do not exceed 30 mg daily of lomitapide when used concomitantly with weak CYP3A4 inhibitors, including atorvastatin and oral contraceptives.
- Increases plasma concentration of warfarin; monitor INR regularly, especially with lomitapide dose adjustment.
- Increased systemic exposure to simvastatin and lovastatin exposure with lomitapide. Limit statin dose when coadministered due to myopathy risk.
- Consider dose reduction of P-glycoprotein substrates because of possible increased absorption with lomitapide.
- Separate lomitapide dosing with BAS by at least 4 hours.
- CV outcomes trials: The effect of lomitapide on CV morbidity and mortality has not been determined
- Considerations in prescribing: Cost, oral administration, requires strict adherence to low-fat diet and gradual dose escalation to reduce GI side effects, requires daily doses of specific vitamins (Vitamin E 400 IU, linoleic acid ≥200 mg, alpha-linolenic acid ≥210 mg, eicosapentaenoic acid ≥110 mg, docosahexaenoic acid ≥80 mg); requires monitoring of transaminase levels, long-term consequences of hepatic steatosis unknown, prescriber training, REMS program
LDL apheresis
- Mechanism of action: Selectively removes apo B-containing lipoproteins, producing an acute reduction in LDL-C.
- FDA approved indication: Patients with FH unresponsive to pharmacologic and dietary management who are either functional homozygotes with an LDL-C >500mg/dL, functional heterozygotes with no known CV disease but an LDL-C >300mg/dL, or functional heterozygotes with known cardiovascular disease and LDL-C >200mg/dL
- Dose and route of administration: Extracorporeal technique performed weekly or biweekly
- Mean % LDL-C reduction: With weekly or biweekly treatment, average LDL-C can ↓ to ~50–60% of the original levels. LDL-C increases after each apheresis session but does not return to the original level
- Adverse effects: Problems with venous access; transient hypotension, fatigue; bleeding; hypocalcemia; iron deficiency due to regular phlebotomy for diagnostic purposes; heparin allergy; and bradykinin syndrome (especially with ACEi)
- Drug–drug interactions: ACEi should not be used with dextran sulfate method owing to risk of bradykinin syndrome
- CV outcomes trials: Limited due to ethical considerations in RCTs of very high-risk patients with HoFH, but it is reasonable to assume reductions in CV disease events are proportional to the degree of LDL-C lowering
- Considerations in prescribing: Cost, extracorporeal technique, inconvenient, locations not readily available in some regions, time-consuming, robust reduction in LDL-C
- ↑ = increase
- ↓ = decrease
- ACEi = angiotensin-converting enzyme inhibitor
- ACL = adenosine triphosphate-citrate lyase
- ACS = acute coronary syndrome
- ACSVl1 = acyl-CoA synthetase-1
- ALT = alanine transaminase
- apoB = apolipoprotein B-100
- ASCVD = atherosclerotic cardiovascular disease
- ANGPTL3 = Angiopoietin-like 3
- AST = aspartate aminotransferase
- BAS = bile acid sequestrant
- CANTAB = Cambridge Neuropsychological Test Automated Battery
- CHD = coronary heart disease
- CKD = chronic kidney disease
- CLEAR Outcomes = Evaluation of Major Cardiovascular Events in Patients With, or at High Risk for, Cardiovascular Disease Who Are Statin Intolerant With Bempedoic Acid [ETC-1002] or Placebo
- CV = cardiovascular
- CYP3A4 = Cytochrome P450 3A4
- EBBINGHAUS = Evaluating PCSK9 Binding antiBody Influence oN coGnitive HeAlth in High cardiovascUlar Risk Subjects
- FDA = Food and Drug Administration
- FH = familial hypercholesterolemia
- FOURIER = Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk
- GI = gastrointestinal
- HbA1c = glycosylated hemoglobin
- HDL-C = high-density lipoprotein cholesterol
- HeFH = heterozygous familial hypercholesterolemia
- HoFH = homozygous familial hypercholesterolemia
- IMPROVE-IT = IMProved Reduction of Outcomes: Vytorin Efficacy International Tria
- IV = intravenous
- LDL-C = low-density lipoprotein cholesterol
- LRC-CPPT = Lipid Research Clinics Coronary Primary Prevention Trial
- mAb = monoclonal antibody
- MI = myocardial infarction
- NPC1L1 = Niemann-Pick C1 like 1
- ODYSSEY = Evaluation of Cardiovascular Outcomes After an Acute Coronary Syndrome During Treatment With Alirocumab
- ORION-4 = A Randomized Trial Assessing the Effects of Inclisiran on Clinical Outcomes Among People With Cardiovascular Disease
- PAD = peripheral arterial disease
- PCSK9 = proprotein convertase subtilisin/kexin type 9
- PI = prescribing information
- RD/RDN = registered dietician/registered dietician nutritionist
- REMS = Risk Evaluation and Mitigation Strategy
- SC = subcutaneous
- SHARP = Study of Heart and Renal Protection
- siRNA = synthetic small interfering ribonucleic acid
- TC = total cholesterol
- TG = triglycerides
- UA = unstable angina
- VICTORION-2P = A Randomized, Double-blind, Placebo-controlled, Multicenter Trial, Assessing the Impact of Inclisiran on Major Adverse Cardiovascular Events in Participants With Established Cardiovascular Disease
- VLDL = very low-density lipoprotein
Secondary Prevention - Patients with Clinical ASCVD
Familial Hypercholesterolemia (FH) Diagnostic Categories
| ICD-10 Category | Clinical Criteria | With Genetic Testing Performed |
| Heterozygous FH |
|
|
| Homozygous FH |
|
|
| Family history of FH |
|
|
- apoB = apolipoprotein B
- CAD = coronary artery disease
- FH = familial hypercholesterolemia
- ICD-10 = International Classification of Disease, 10th Revision
- LDL = low-3 density lipoprotein
- LDL-C = low-density lipoprotein cholesterol
- PCSK9 = proprotein convertase subtilisin/kexin type 9
External Links & References
Hypertriglyceridemia Key Definitions and Assumptions
| Persistent Hypertriglyceridemia | Fasting triglycerides > 150 mg/dL following a minimum of 4 to 12 weeks of lifestyle intervention, a stable dose of maximally tolerated statin therapy when indicated, as well as evaluation and management of secondary causes of hypertriglyceridemia. Before beginning triglyceride risk-based nonstatin therapies, a fasting lipid panel should be obtained (2 measurements of fasting lipids, preferably at least 2 weeks apart is recommended). |
| Fasting vs Nonfasting Lipid Measurement | In most patients, the postprandial rise in triglycerides is small, between 12 and 27 mg/dL. The 2018 AHA/ACC/ multisociety cholesterol guideline recommends that for adults aged > 20 years not taking lipid-lowering drug therapy, either a fasting or nonfasting lipid profile may be used to estimate ASCVD risk and document baseline LDL-C. For those with nonfasting triglycerides > 400 mg/dL, a repeat fasting lipid profile is recommended to assess fasting triglycerides and baseline LDL-C. The Martin-Hopkins method provides accurate assessments of LDL-C in individuals with hypertriglyceridemia. Fasting lipid testing is favored under the following circumstances: a) To establish the diagnosis of the metabolic syndrome, as one of the criteria is fasting triglycerides > 150 mg/dL b) To identify lipid disorders in those without ASCVD, but with a family history of premature ASCVD or genetic lipid disorders c) To assess adherence to lifestyle and medical therapy in those being treated with lipid-lowering medication d) To identify those with triglycerides > 500 mg/dL, who are at risk for hypertriglyceridemia-induced pancreatitis, and monitor their response to therapy. |
| Secondary Causes of Hypertriglyceridemia | It is crucial that clinicians investigate and treat secondary causes of hypertriglyceridemia. Diseases, diet/lifestyle, medications, and disorders of metabolism are major causes for elevation of triglycerides that clinicians can use to rule out secondary causes of hypertriglyceridemia. These factors can either cause or contribute to triglyceride elevations in patients. Poor glycemic control may significantly influence plasma lipid levels in patients with diabetes mellitus and significantly exacerbate hypertriglyceridemia. A genetic predisposition to hypertriglyceridemia increases the likelihood and severity of elevated triglycerides in each category. Multifactorial chylomicronemia syndrome is the most common condition that elevates triglyceride levels high enough to cause lipemia retinalis, eruptive xanthomas, abdominal pain, and hyperlipidemic pancreatitis. Clinicians should understand the drugs and conditions which make this disease more likely as pancreatitis associated with hypertriglyceridemia can be fatal. |
| Lifestyle Intervention | Lifestyle modification (ie, adherence to a heart-healthy diet, regular physical activity, avoidance of tobacco products, limited alcohol consumption, and maintenance of a healthy weight) remains a critical component of ASCVD risk reduction, both before and in concert with the use of lipid-lowering medications. Referral to a registered dietitian nutritionist is strongly recommended to improve understanding of heart-healthy dietary principles and individualize nutrition recommendations for patients with hypertriglyceridemia. Given that metabolic risk factors such as hypertriglyceridemia cluster with other metabolic risk factors (abdominal obesity, hypertension, hyperglycemia), adherence to a recommended dietary intervention can markedly benefit the entire metabolic risk profile over the life course. Adherence to lifestyle modification should be regularly assessed at the time of initiation or modification of statin therapy and at each patient visit during monitoring of ongoing therapy. |
| Role of Statin Therapy in Patients with Hypertriglyceridemia | Although commonly recognized for their impact on LDL-C, statins also provide a 10 to 30% dose-dependent triglyceride reduction in patients with elevated triglyceride levels. Trials have demonstrated those with elevated triglyceride levels are at increased risk of ASCVD events and can achieve ASCVD risk reduction with statin therapy. |
| Persistent Hypertriglyceridemia as a Risk-Enhancing Factor in Primary Prevention | Persistently elevated triglycerides (nonfasting triglycerides > 175 mg/dL) are one of the risk-enhancing factors identified by the 2018 AHA/ACC/multisociety cholesterol guideline, according to which the 10-year ASCVD risk derived using the Pooled Cohort Equations (PCE) is a useful tool to predict population risk. However, clinicians should be aware that it has limitations when applied to individuals. The PCE may overestimate risk in individuals from higher socioeconomic status, as well as in those receiving consistent screening and preventive care. One purpose of the discussion is to individualize risk status based on the PCE estimate as well as other factors. These factors may suggest a higher lifetime risk than is denoted by the 10-year risk estimate with the PCE. |
| Role of Omega-3 Fatty Acids in Patients with Hypertriglyceridemia |
|
Class of Recommendation and Level of Evidence Table
Applying the American College of Cardiology/American Heart Association Class of Recommendation and Level of Evidence to Clinical Strategies, Interventions, Treatments, or Diagnostic Testing in Patient Care* (Updated December 2024)Recommendations are designated with both a class of recommendation (COR) and a level of evidence (LOE). The class of recommendation indicates the strength of recommendation, encompassing the estimated magnitude and certainty of benefit in proportion to risk. The level of evidence rates the quality of scientific evidence supporting the intervention on the basis of the type, quantity, and consistency of data from clinical trials and other sources.
- Is recommended
- Is indicated/useful/effective/beneficial
- Should be performed/administered/other
-
Comparative-Effectiveness Phrases†:
- Treatment/strategy A is recommended/indicated in preference to treatment B
- Treatment A should be chosen over treatment B
- Is reasonable
- Can be useful/effective/beneficial
-
Comparative-Effectiveness Phrases†:
- Treatment/strategy A is probably recommended/indicated in preference to treatment B
- It is reasonable to choose treatment A over treatment B
- May/might be reasonable
- May/might be considered
- Usefulness/effectiveness is unknown/unclear/uncertain or not well-established
- Is not recommended
- Is not indicated/useful/effective/beneficial
- Should not be performed/administered/other
- Potentially harmful
- Causes harm
- Associated with excess morbidity/mortality
- Should not be performed/administered/other
LEVEL (QUALITY) OF EVIDENCEǂ
- High-quality evidenceǂ from more than 1 RCT
- Meta-analyses of high-quality RCTs
- One or more RCTs corroborated by high-quality registry studies
- Moderate-quality evidenceǂ from 1 or more RCTs
- Meta-analyses of moderate-quality RCTs
- Moderate-quality evidenceǂ from 1 or more well-designed, well-executed nonrandomized studies, observational studies, or registry studies
- Meta-analyses of such studies
- Randomized or nonrandomized observational or registry studies with limitations of design or execution
- Meta-analyses of such studies
- Physiological or mechanistic studies in human subjects
- Consensus of expert opinion based on clinical experience
COR and LOE are determined independently (any COR may be paired with any LOE).
A recommendation with LOE C does not imply that the recommendation is weak. Many important clinical questions addressed in guidelines do not lend themselves to clinical trials. Although RCTs are unavailable, there may be a very clear clinical consensus that a particular test or therapy is useful or effective.
*The outcome or result of the intervention should be specified (an improved clinical outcome or increased diagnostic accuracy or incremental prognostic information).
† For comparative-effectiveness recommendations (COR 1 and 2a; LOE A and B only), studies that support the use of comparator verbs should involve direct comparisons of the treatments or strategies being evaluated.
‡ The method of assessing quality is evolving, including the application of standardized, widely used, and preferably validated evidence grading tools; and for systematic reviews, the incorporation of an Evidence Review Committee.
COR indicates Class of Recommendation; EO, expert opinion; LD, limited data; LOE, Level of Evidence; NR, nonrandomized; R, randomized; and RCT, randomized controlled trial.
Lipoprotein Goals for ASCVD Risk Reduction
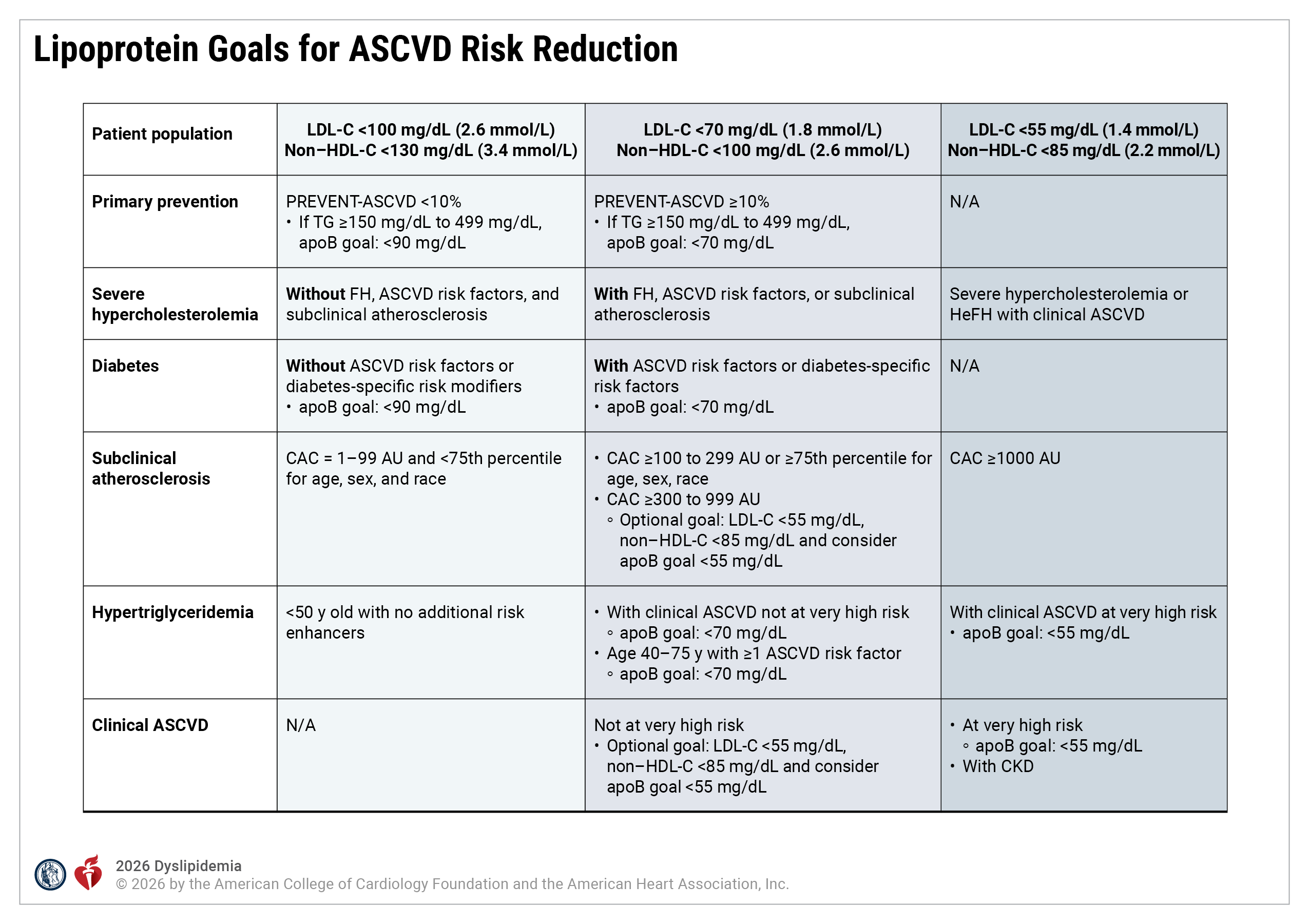
apoB indicates apolipoprotein B; ASCVD, atherosclerotic cardiovascular disease; AU, Agatston units; CAC, coronary artery calcium; CKD, chronic kidney disease; FH, familial hypercholesterolemia; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; and TG, triglycerides.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 1CPR Framework for Risk Evaluation
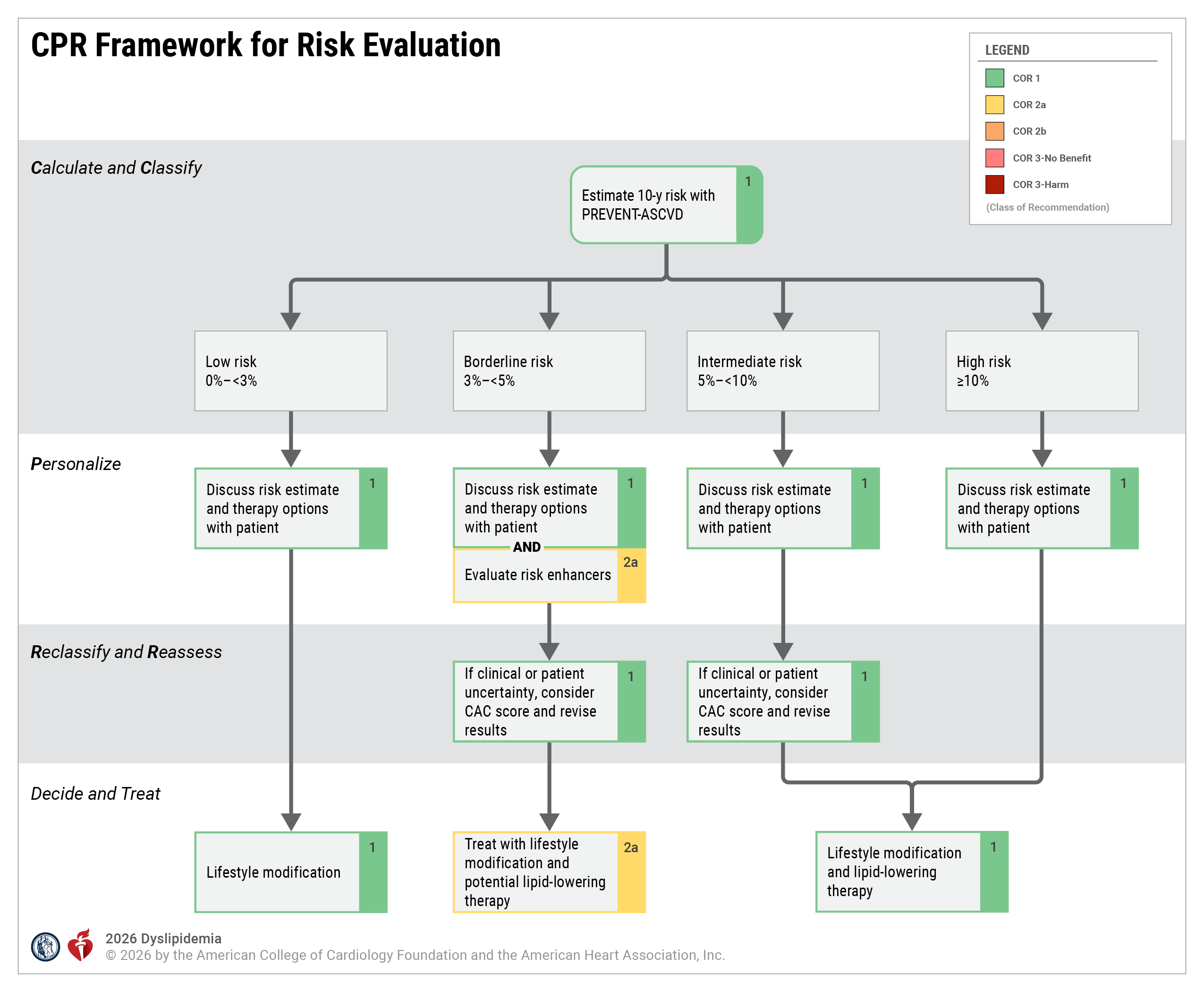
CAC indicates coronary artery calcium; and CPR, Calculate–Personalize–Reclassify.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 5Intensities of Statin Therapy
High-, Moderate-, and Low-Intensity Statin Therapy*
| High-Intensity | Moderate-Intensity | Low-Intensity | |
|---|---|---|---|
| Expected % LDL-C Reduction † |
≥ 50% | 30% to 49% | < 30% |
| Preferred Statins |
Atorvastatin (40 mg) 80 mg Rosuvastatin 20 mg (40 mg) |
Atorvastatin 10 mg (20 mg) Rosuvastatin (5 mg) 10 mg |
|
| Other Statins |
- |
Fluvastatin XL 80 mg Fluvastatin 40 mg BID Lovastatin 40 mg (80 mg) Pitavastatin 1, 2, 4 mg Pravastatin 40 mg (80 mg) Simvastatin 20, 40 mg‡ |
Fluvastatin 20, 40 mg Lovastatin 20 mg Pravastatin 10, 20 mg Simvastatin 10 mg |
Expected percentage LDL-C reductions with atorvastatin, rosuvastatin, and simvastatin were estimated using the median reduction in LDL-C from the VOYAGER database. Reductions in LDL-C for other statins (fluvastatin, lovastatin, pitavastatin, and pravastatin) were identified according to FDA-approved product labeling in adults with hyperlipidemia, primary hypercholesterolemia, and mixed dyslipidemia. Boldface type indicates specific statins and doses that were evaluated in placebo-controlled RCTs evaluating ASCVD event lowering, and the Cholesterol Treatment Trialists’ 2010 meta-analysis. These RCTs demonstrated a reduction in major ASCVD events. Modified with permission from Grundy et al. Copyright 2018 American Heart Association, Inc. and American College of Cardiology Foundation.
*Expected percentage reductions are estimates from data across large populations. Individual responses to statin therapy varied in the RCTs and should be expected to vary in clinical practice owing to a high degree of heterogeneity seen with LDL-C–lowering medications.
†Expected LDL-C lowering with the dosage listed below each intensity.
‡Although simvastatin 80 mg was evaluated in RCTs, initiation of simvastatin 80 mg or titration to 80 mg is not recommended by the FDA because of the increased risk of myopathy, including rhabdomyolysis.
BID indicates twice daily; FDA, U.S. Food and Drug Administration; LDL-C, low-density lipoprotein-cholesterol; RCT, randomized controlled trial; and XL, extended release.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 6Characteristics of Common Lipid-Lowering Medications to Treat Dyslipidemia*
Class: HMG-CoA reductase inhibitors (aka statins)
Mechanism of Action: Competitively inhibits HMG-CoA reductase (rate-limiting step of endogenous cholesterol production); increases LDL receptor expression on the surface of hepatocytes
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Atorvastatin | 10-80 mg | Once daily |
| Fluvastatin | 20-80 mg | Once or twice daily |
| Lovastatin | 10-80 mg | Once or twice daily |
| Pitavastatin | 1-4 mg | Once daily |
| Pravastatin | 10-80 mg | Once daily |
| Rosuvastatin | 5-40 mg | Once daily |
| Simvastatin | 5-40 mg | Once daily |
Guideline Comments†:
- Oral agents
- First-line therapy for ASCVD risk reduction in nearly all patients
- Extensive CVOT evidence demonstrates reductions in events over wide range of LDL-C levels in primary and secondary prevention
- Expected LDL-C reduction: 18%-55%‡
- Atorvastatin and rosuvastatin can achieve high-intensity LDL-C reductions and are preferred for patients at high/very high ASCVD risk
- Atorvastatin, pitavastatin, and rosuvastatin have long half-lives that enable dosing any time of the day
Common Side Effects:
- Myalgia (normal CK level) is common and dose-related
Contraindications‖:
- Acute liver failure
- Decompensated cirrhosis
- Lactation
- Severe underlying neuromuscular diseases (eg, dermatomyositis, muscular dystrophy)
Comments:
- See 2026 Dyslipidemia Guideline Section 4.2.11, “Management of Statin-Attributed Muscle Symptoms”
- Myositis/myopathy (CK >ULN) with concerning symptoms or objective weakness is rare; rhabdomyolysis (CK >10 times ULN with renal injury) is rare; immune-mediated necrotizing myopathy (with HMGCR antibodies) is very rare
- Transaminase elevations (>3 times ULN) are rare
- Postmarketing reports of cognitive impairment (eg, memory loss, forgetfulness) are rare, generally nonserious, reversible upon statin discontinuation, and not observed in prospective clinical trials
- See 2026 Dyslipidemia Guideline Section 4.2.8.4, “Management of Dyslipidemia in Persons Planning Pregnancy, During Pregnancy, or While Lactating,” pregnancy is not a contraindication, but statins should be avoided while lactating (the Food and Drug Administration has removed pregnancy as a contraindication to statin therapy)
FDA-approved indication(s)§:
FDA labels are available on FDA website:
Class: Cholesterol absorption inhibitor
Mechanism of Action: Blocks the sterol transporter protein, NPC1L1, to inhibit intestinal and biliary sterol absorption; increases LDL receptor expression on the surface of hepatocytes secondary to reduced hepatic sterol levels
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Ezetimibe | 10 mg | Once daily |
Guideline Comments†:
- Oral agents
- CVOT evidence demonstrates reductions in cardiovascular events in very high-risk secondary prevention in combination with a moderate-intensity statin therapy
- Expected LDL-C reduction: monotherapy, 18%; combination with a statin, 25% incremental reduction‡
- Drug of choice in sitosterolemia to reduce elevated sitosterol and campesterol
Common Side Effects:
- Typically well tolerated
Contraindications‖:
- Prior hypersensitivity to ezetimibe
Comments:
- Not recommended in patients with moderate or severe hepatic impairment
- Persistent elevations in hepatic transaminase (>3 times ULN) can occur when added to a statin; monitor hepatic transaminase levels before and after initiating statin with ezetimibe combination therapy
FDA-approved indication(s)§:
Ezetimibe:
ZETIA is indicated:
- In combination with a statin, or alone when additional low density lipoprotein cholesterol (LDL-C) lowering therapy is not possible, as an adjunct to diet to reduce elevated LDL-C in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH).
- In combination with a statin as an adjunct to diet to reduce elevated LDL-C in pediatric patients 10 years of age and older with HeFH.
- In combination with fenofibrate as an adjunct to diet to reduce elevated LDL-C in adults with mixed hyperlipidemia.
- In combination with a statin, and other LDL-C lowering therapies, to reduce elevated LDL-C levels in adults and in pediatric patients 10 years of age and older with homozygous familial hypercholesterolemia (HoFH).
- As an adjunct to diet for the reduction of elevated sitosterol and campesterol levels in adults and in pediatric patients 9 years of age and older with homozygous familial sitosterolemia.
When ZETIA is used in combination with a statin, fenofibrate, or other LDL-C lowering therapies, refer to the Prescribing Information of these products for information on the safe and effective use.
§Currently the FDA website only has label information for Zetia, so its FDA indications are presented here.
Class: PCSK9 inhibitor: monoclonal antibodies
Mechanism of Action: Fully human monoclonal antibodies: binds to PCSK9 in the circulation and decreases degradation of LDL receptors
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Alirocumab | 75-150 mg, | Every 2 weeks |
| or 300 mg | Every 4 weeks | |
| Evolocumab | 140 mg | Every 2 weeks |
Guideline Comments†:
- Subcutaneous agents
- CVOT evidence demonstrates reductions in cardiovascular events in very high-risk secondary prevention in combination with a maximally tolerated statin therapy
- Expected LDL-C reduction: 45%-64%‡
- Lower mean LDL-C reduction (21%-31%) in homozygous FH due to LDLR gene variants
Common Side Effects:
- Injection site reactions
Contraindications‖:
- Prior hypersensitivity to the PCSK9 monoclonal antibody
Comments:
- Hypersensitivity reactions (including angioedema) are rare
- Latex is in some evolocumab single-dose prefilled syringe covers; alirocumab products do not contain latex
FDA-approved indication(s)§:
Alirocumab:
PRALUENT is a proprotein convertase subtilisin kexin type 9 (PCSK9) inhibitor indicated:
- To reduce the risk of major adverse cardiovascular (CV) events (coronary heart disease death, myocardial infarction, stroke, or unstable angina requiring hospitalization) in adults at increased risk for these events.
- As an adjunct to diet and exercise to reduce low-density lipoprotein cholesterol (LDL-C) in:
- adults with hypercholesterolemia.
- adults and pediatric patients aged 8 years and older with heterozygous familial hypercholesterolemia (HeFH).
- adults with homozygous familial hypercholesterolemia (HoFH).
Evolocumab:
REPATHA is a PCSK9 (proprotein convertase subtilisin kexin type 9) inhibitor indicated:
- To reduce the risk of major adverse cardiovascular (CV) events (CV death, myocardial infarction, stroke, unstable angina requiring hospitalization, or coronary revascularization) in adults at increased risk for these events.
- As an adjunct to diet and exercise to reduce low-density lipoprotein cholesterol (LDL-C) in:
- adults with hypercholesterolemia.
- adults and pediatric patients aged 10 years and older with heterozygous familial hypercholesterolemia (HeFH).
- adults and pediatric patients aged 10 years and older with homozygous familial hypercholesterolemia (HoFH).
§Currently the FDA website only has label information for Praluent and Repatha, so their FDA indications are presented here.
Class: ATP citrate lyase inhibitor
Mechanism of Action: Inhibits ATP citrate lyase in the liver to decrease cholesterol production upstream of HMG-Co reductase in the cholesterol synthesis pathway; increases LDL receptor expression on the surface of hepatocytes
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Bempedoic acid | 180 mg | Once daily |
Guideline Comments†:
- Oral agents
- CVOT evidence demonstrating reductions in cardiovascular events in individuals treated for high-risk primary and secondary prevention with statin-attributed side effects
- Prodrug that is activated by very long-chain acyl-CoA synthetase, found primarily in the liver
- Expected LDL-C reduction: monotherapy in patients with statin-attributed side effects, 21%-24% combination with a statin, 17%-18%‡
Common Side Effects:
- Typically well tolerated
Contraindications‖:
- Prior serious hypersensitivity reaction to bempedoic acid or any of the excipients
Comments:
- Elevated BUN, creatinine, and uric acid have been reported (monitoring serum urate level necessary in patients with preexisting and untreated hyperuricemia)
FDA-approved indication(s)§:
Bempedoic Acid:
NEXLETOL, an adenosine triphosphate-citrate lyase (ACL) inhibitor, is indicated:
- to reduce the risk of major adverse cardiovascular events (cardiovascular death, myocardial infarction, stroke, or coronary revascularization) in adults at increased risk for these events who are unable to take recommended statin therapy (including those not taking a statin).
- as an adjunct to diet and exercise, in combination with other low-density lipoprotein cholesterol (LDL-C) lowering therapies, or alone when concomitant LDL-C lowering therapy is not possible, to reduce LDL-C in adults with hypercholesterolemia, including heterozygous familial hypercholesterolemia (HeFH).
Bempedoic Acid and Ezetimibe (combination therapy):
NEXLIZET, a combination of bempedoic acid, an adenosine triphosphate citrate lyase (ACL) inhibitor, and ezetimibe, a dietary cholesterol absorption inhibitor, is indicated:
- As an adjunct to diet and exercise to reduce low-density lipoprotein cholesterol (LDL-C) in adults with hypercholesterolemia, including heterozygous familial hypercholesterolemia (HeFH).
§Currently Nexletol is the only bempedoic acid and Nexlizet is the only bempedoic acid combination therapy, so their FDA indications are presented here.
Class: PCSK9 inhibitor: small interfering RNA
Mechanism of Action: Utilizes endogenous RNA interference mechanism and directs catalytic breakdown of mRNA for PCSK9 and decreases degradation of the LDL receptor
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Inclisiran | 284 mg | Initial dose followed by second dose in 3 months, then every 6 months thereafter |
Guideline Comments†:
- Subcutaneous agent administered by a health care professional
- Expected LDL-C reduction: 48%-52%‡
- CVOT in progress
Common Side Effects:
- Injection site reactions
Contraindications‖:
- Prior serious hypersensitivity to inclisiran or any of the excipients
Comments:
- Hypersensitivity reactions (including angioedema) are rare
FDA-approved indication(s)§:
Inclisiran:
LEQVIO is a small interfering RNA (siRNA) directed to proprotein convertase subtilisin kexin type 9 (PCSK9) mRNA indicated as an adjunct to diet and exercise to reduce low-density lipoprotein cholesterol (LDL-C) in:
- adults with hypercholesterolemia.
- adults and pediatric patients aged 12 years and older with heterozygous familial hypercholesterolemia (HeFH).
- pediatric patients aged 12 years and older with homozygous familial hypercholesterolemia (HoFH).
§Currently the FDA website only has label information for Leqvio, so its FDA indications are presented here.
Class: Bile acid sequestrants
Mechanism of Action: Bind bile acids in the gut, interrupt enterohepatic recirculation of bile acids and impede their reabsorption, decrease bile acid pooling in the liver, increase conversion of cholesterol to bile acids; increase LDL receptor expression on the surface of hepatocytes
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Cholestyramine | 8-16 g | Once or twice daily |
| Colesevelam | 3.75 mg | Once or twice daily |
| Colestipol | 2-16 g | Once or multiple times daily |
Guideline Comments†:
- CVOT evidence from 1 trial demonstrating cardiovascular event reduction in primary prevention men as monotherapy
- Nonsystemic, add-on to statin therapy or in patients with statin-intolerance
- Expected LDL-C reduction: 10%-27%‡
- Gastrointestinal side effects may limit use
- Both LDL-C lowering and incidence of side effects are dose related
- May increase serum TG levels; avoid if TG ≥300 mg/dL
Common Side Effects:
- Abdominal pain
- Bloating
- Dyspepsia
- Nausea
- Constipation
Contraindications‖:
- TG >500 mg/dL (colesevelam)
- History of hypertriglyceridemia-induced pancreatitis
- Bowel obstruction
Comments:
- Poor tolerability due to side effects and potential drug–drug interactions limits use; may decrease absorption of fat-soluble vitamins and folic acid; generally considered the safest medication in pregnancy
FDA-approved indication(s)§:
Cholestyramine:
1) Cholestyramine for oral suspension, USP powder is indicated as adjunctive therapy to diet for the reduction of elevated serum cholesterol in patients with primary hypercholesterolemia (elevated low density lipoprotein [LDL] cholesterol) who do not respond adequately to diet. Cholestyramine for oral suspension, USP powder may be useful to lower LDL cholesterol in patients who also have hypertriglyceridemia, but it is not indicated where hypertriglyceridemia is the abnormality of most concern.
Therapy with lipid-altering agents should be a component of multiple risk factor intervention in those individuals at significantly increased risk for atherosclerotic vascular disease due to hypercholesterolemia. Treatment should begin and continue with dietary therapy specific for the type of hyperlipoproteinemia determined prior to initiation of drug therapy. Excess body weight may be an important factor and caloric restriction for weight normalization should be addressed prior to drug therapy in the overweight.
Prior to initiating therapy with cholestyramine resin, secondary causes of hypercholesterolemia (e.g., poorly
controlled diabetes mellitus, hypothyroidism, nephrotic syndrome, dysproteinemias, obstructive liver disease,
other
drug therapy, alcoholism), should be excluded and a lipid profile performed to assess Total cholesterol, HDL-C and
triglycerides (TG). For individuals with TG less than 400 mg/dL (<4.5 mmol/L), LDL-C can be estimated using the
following equation:
LDL-C = Total cholesterol - [(TG/5) + HDL-C]
For TG levels > 400 mg/dL, this equation is less accurate and LDL-C concentrations should be determined by ultracentrifugation. In hypertriglyceridemic patients, LDL-C may be low or normal despite elevated Total-C. In such cases cholestyramine resin may not be indicated.
Serum cholesterol and triglyceride levels should be determined periodically based on NCEP guidelines to confirm initial and adequate long-term response. A favorable trend in cholesterol reduction should occur during the first month of cholestyramine resin therapy. The therapy should be continued to sustain cholesterol reduction. If adequate cholesterol reduction is not attained, increasing the dosage of cholestyramine resin or adding other lipid-lowering agents in combination with cholestyramine resin should be considered.
Since the goal of treatment is to lower LDL-C, the NCEP recommends that LDL-C levels be used to initiate and assess treatment response. If LDL-C levels are not available then Total-C alone may be used to monitor long-term therapy. A lipoprotein analysis (including LDL-C determination) should be carried out once a year. The NCEP treatment guidelines are available on the FDA website.
Cholestyramine resin monotherapy has been demonstrated to retard the rate of progression and increase the rate of regression of coronary atherosclerosis.
2) Cholestyramine for oral suspension, USP powder is indicated for the relief of pruritus associated with partial biliary obstruction. Cholestyramine resin has been shown to have a variable effect on serum cholesterol in these patients. Patients with primary biliary cirrhosis may exhibit an elevated cholesterol as part of their disease.
FDA Label information for Cholestyramine:
https://www.accessdata.fda.gov/drugsatfda_docs/nda/97/74771_Cholestyramine_prntlbl.pdf
Colesevelam:
WELCHOL is a bile acid sequestrant indicated as an adjunct to diet and exercise to:
- reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia.
- reduce LDL-C levels in boys and postmenarchal girls, 10 to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH), unable to reach LDL-C target levels despite an adequate trial of diet and lifestyle modification.
- improve glycemic control in adults with type 2 diabetes mellitus.
Limitations of Use:
- Do not use for treatment of type 1 diabetes or for diabetic ketoacidosis.
- Not studied in Fredrickson Type I, III, IV, and V dyslipidemias.
§Currently the FDA website only has label information for Welchol, so its FDA indications are presented here.
Colestipol:
Since no drug is innocuous, strict attention should be paid to the indications and contraindications, particularly when selecting drugs for chronic long-term use.
COLESTID Tablets are indicated as adjunctive therapy to diet for the reduction of elevated serum total and LDL-C in patients with primary hypercholesterolemia (elevated LDL-C) who do not respond adequately to diet. Generally, COLESTID Tablets have no clinically significant effect on serum triglycerides, but with their use, triglyceride levels may be raised in some patients.
Therapy with lipid-altering agents should be a component of multiple risk factor intervention in those individuals at significantly increased risk for atherosclerotic vascular disease due to hypercholesterolemia. Treatment should begin and continue with dietary therapy (see NCEP guidelines). A minimum of six months of intensive dietary therapy and counseling should be carried out prior to initiation of drug therapy. Shorter periods may be considered in patients with severe elevations of LDL-C or with definite CHD.
According to the NCEP guidelines, the goal of treatment is to lower LDL-C, and LDL-C is to be used to initiate and assess treatment response. Only if LDL-C levels are not available, should the Total-C be used to monitor therapy. The NCEP treatment guidelines are available on the FDA website.
§Currently the FDA website only has label information for Colestid, so its FDA indications are presented here.
Class: Microsomal TG transfer protein inhibitor
Mechanism of Action: Binds and inhibits microsomal TG transfer protein, which is essential for the assembly of apoB-containing lipoproteins, and inhibiting synthesis of chylomicrons and VLDL, which lowers LDL-C
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Lomitapide | 5-60 mg | Once daily |
Guideline Comments†:
- Oral agents
- Expected LDL-C reduction: 40%-50%‡
- Only available through a restricted program; several adverse reactions (including hepatotoxicity due to hepatic steatosis exacerbated by concomitant ethanol intake, as well as steatorrhea) and drug-drug interactions
- Daily vitamin E, linoleic acid, alphalinolenic acid, EPA, and DHA supplements are needed to mitigate reduced absorption of fat-soluble vitamins/fatty acids
- Multiple potential drug interactions associated with CYP3A4 metabolism
Common Side Effects:
- Gastrointestinal effects (ie, diarrhea, nausea, vomiting, dyspepsia, abdominal pain) are common
- Elevated hepatic transaminases
- Increased hepatic fat
- Embryo-fetal toxicity
Contraindications‖:
- Pregnancy
- Concomitant use with a CPY3A4 inhibitor
- Moderate or severe hepatic impairment or active liver disease including unexplained persistent abnormal liver function tests
Comments:
- Use is restricted to patients with HoFH
- Available only through a restricted REMS program that ensures safe use requiring regular monitoring of liver function and other health parameters
FDA-approved indication(s)§:
Lomitapide:
JUXTAPID is a microsomal triglyceride transfer protein inhibitor indicated as an adjunct to a low-fat diet and exercise and other low-density lipoprotein cholesterol (LDL-C) therapies, to reduce LDL-C in adult and pediatric patients aged 2 years and older with HoFH.
§Currently the FDA website only has label information for Juxtapid, so its FDA indications are presented here.
Class: ANGPTL3 inhibitor
Mechanism of Action: Fully human monoclonal antibody that binds to and inhibits ANGPTL3; ANGPTL3 inhibition enhances remnant lipoprotein clearance, reducing VLDL remnants and their conversion into LDL particles
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Evinacumab-dgnb | 15 mg/kg | every 4 weeks |
Guideline Comments†:
- Intravenously administered agent
- Works through an LDL receptor-independent pathway
- Expected LDL-C reduction: 49%‡
Common Side Effects:
- Serious hypersensitivity reactions
- Embryo-fetal toxicity
Contraindications‖:
- History of serious hypersensitivity reactions to evinacumab-dgnb or to any of its excipients
Comments:
- Use is restricted to patients with HoFH
- Infusion rate can be slowed, interrupted, or discontinued if signs of adverse reactions, including infusion or hypersensitivity reactions are present during administration
FDA-approved indication(s)§:
Evinacumab:
EVKEEZA is indicated as an adjunct to diet and exercise and other low-density lipoprotein-cholesterol (LDL-C) lowering therapies to reduce LDL-C in adults and pediatric patients, aged 1 year and older, with homozygous familial hypercholesterolemia (HoFH).
§Currently the FDA website only has label information for Evkeeza, so its FDA indications are presented here.
Class: Fibrates
Mechanism of Action: Stimulates PPAR-alpha, which activates lipoprotein lipase and reduces apolipoprotein C-III production; increases lipolysis and elimination of TG-rich particles
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Fenofibrate | 40-200 mg | Once daily |
| Fenofibric acid | 35-135 mg | Once daily |
| Gemfibrozil | 600 mg | Twice daily |
Guideline Comments†:
- Oral agents
- CVOTs show no reduction in cardiovascular events when fenofibrate is added to statin therapy in primary prevention patients with diabetes; CVOT evidence demonstrating reductions in cardiovascular events in primary and secondary prevention patients with gemfibrozil monotherapy
- First-line option for severe hypertriglyceridemia (≥500 mg/dL, especially when ≥1000 mg/dL)
- Expected TG reduction: 30%-50%‡
- Many different formulations of fenofibrate and fenofibric acid are available with varied dosages
- Dose may need to be reduced in decreased kidney function; avoid in severe kidney dysfunction
- Blunted to no TG reduction in patients with familial chylomicronemia syndrome due to reduced lipoprotein lipase activity
- Gemfibrozil should not be combined with statin therapy because of serious potential drug interaction
Common Side Effects:
- Myalgia
- Persistently elevated hepatic transaminases
Contraindications‖:
- Severe renal dysfunction
- Active liver disease
- Gallbladder disease
- During lactation
- History of hypersensitivity to the fibrate product
Comments:
- Fenofibrate and fenofibric acid can reversibly elevate serum creatinine
- When given in combination with statin therapy can increase risk of muscle toxicity; gemfibrozil should not be used in combination with a statin; fenofibrate or fenofibric acid can be used in combination with statin with monitoring
FDA-approved indication(s)§:
Fenofibrate:
Antara is a peroxisome proliferator-activated receptor (PPAR) alpha agonist indicated as an adjunct to diet:
- to reduce triglyceride (TG) levels in adults with severe hypertriglyceridemia (TG greater than or equal to 500 mg/dL).
- to reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia when use of recommended LDL-C lowering therapy is not possible.
Limitations of Use:
- Markedly elevated levels of serum TG (e.g., > 2,000 mg/dL) may increase the risk of developing pancreatitis. The effect of fenofibrate therapy on reducing this risk has not been determined.
- Fenofibrate did not reduce coronary heart disease morbidity and mortality in two large, randomized controlled trials of patients with type 2 diabetes mellitus.
§Currently the FDA website only has label information for Antara, so its FDA indications are presented here.
Fenofibric acid:
Trilipix is a peroxisome proliferator-activated receptor (PPAR) alpha agonist indicated as adjunct to diet:
- to reduce triglyceride (TG) levels in adults with severe hypertriglyceridemia (TG greater than or equal to 500 mg/dL).
- to reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia when use of recommended LDL-C lowering therapy is not possible.
Limitations of Use:
- Markedly elevated levels of serum TG (e.g., >2,000 mg/dL) may increase the risk of developing pancreatitis. The effect of fenofibrate therapy on reducing this risk has not been determined.
- Fenofibrate did not reduce coronary heart disease morbidity and mortality in two large, randomized controlled trials of patients with type 2 diabetes mellitus.
§Currently the FDA website only has label information for Trilipix, so its FDA indications are presented here.
Gemfibrozil:
LOPID (gemfibrozil tablets, USP) is indicated as adjunctive therapy to diet for:
1) Treatment of adult patients with very high elevations of serum triglyceride levels (Types IV and V hyperlipidemia) who present a risk of pancreatitis and who do not respond adequately to a determined dietary effort to control them. Patients who present such risk typically have serum triglycerides over 2000 mg/dL and have elevations of VLDL-cholesterol as well as fasting chylomicrons (Type V hyperlipidemia). Subjects who consistently have total serum or plasma triglycerides below 1000 mg/dL are unlikely to present a risk of pancreatitis. LOPID therapy may be considered for those subjects with triglyceride elevations between 1000 and 2000 mg/dL who have a history of pancreatitis or of recurrent abdominal pain typical of pancreatitis. It is recognized that some Type IV patients with triglycerides under 1000 mg/dL may, through dietary or alcoholic indiscretion, convert to a Type V pattern with massive triglyceride elevations accompanying fasting chylomicronemia, but the influence of LOPID therapy on the risk of pancreatitis in such situations has not been adequately studied. Drug therapy is not indicated for patients with Type I hyperlipoproteinemia, who have elevations of chylomicrons and plasma triglycerides, but who have normal levels of very low density lipoprotein (VLDL). Inspection of plasma refrigerated for 14 hours is helpful in distinguishing Types I, IV, and V hyperlipoproteinemia.
2) Reducing the risk of developing coronary heart disease only in Type IIb patients without history of or symptoms of existing coronary heart disease who have had an inadequate response to weight loss, dietary therapy, exercise, and other pharmacologic agents (such as bile acid sequestrants and nicotinic acid, known to reduce LDL- and raise HDL-cholesterol) and who have the following triad of lipid abnormalities: low HDL-cholesterol levels in addition to elevated LDL-cholesterol and elevated triglycerides (see WARNINGS, PRECAUTIONS, and CLINICAL PHARMACOLOGY). The National Cholesterol Education Program has defined a serum HDL-cholesterol value that is consistently below 35 mg/dL as constituting an independent risk factor for coronary heart disease. Patients with significantly elevated triglycerides should be closely observed when treated with gemfibrozil. In some patients with high triglyceride levels, treatment with gemfibrozil is associated with a significant increase in LDL-cholesterol. BECAUSE OF POTENTIAL TOXICITY SUCH AS MALIGNANCY, GALLBLADDER DISEASE, ABDOMINAL PAIN LEADING TO APPENDECTOMY AND OTHER ABDOMINAL SURGERIES, AN INCREASED INCIDENCE IN NON-CORONARY MORTALITY, AND THE 44% RELATIVE INCREASE DURING THE TRIAL PERIOD IN AGE-ADJUSTED ALL-CAUSE MORTALITY SEEN WITH THE CHEMICALLY AND PHARMACOLOGICALLY RELATED DRUG, CLOFIBRATE, THE POTENTIAL BENEFIT OF GEMFIBROZIL IN TREATING TYPE IIA PATIENTS WITH ELEVATIONS OF LDL-CHOLESTEROL ONLY IS NOT LIKELY TO OUTWEIGH THE RISKS. LOPID IS ALSO NOT INDICATED FOR THE TREATMENT OF PATIENTS WITH LOW HDL-CHOLESTEROL AS THEIR ONLY LIPID ABNORMALITY.
§Currently the FDA website only has label information for Lopid, so its FDA indications are presented here.
Class: Omega-3 fatty acids
Mechanism of Action: Reduces hepatic VLDL TG synthesis and/or secretion; enhances TG clearance from circulating VLDL; other mechanisms are possible
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Icosapent ethyl | 4 g | Twice daily with food |
| Omega-3 acid ethyl esters | 4 g | Once or twice daily with food |
Guideline Comments†:
- Oral agents
- First-line option for severe (≥500 mg/dL) hypertriglyceridemia
- CVOT evidence demonstrating reductions in cardiovascular events with icosapent ethyl in individuals being treated for primary prevention of ASCVD patients with type 2 diabetes or secondary prevention if TG between 150-499 mg/dL and LDL-C is 41-100 mg/dL despite statin therapy
- Expected TG reduction: 15%-61%‡
- Icosapent ethyl contains EPA only
- Omega-3 acid ethyl esters contain DHA and EPA, which can modestly raise LDL-C (but not apoB)
- Nonprescription fish oil supplements are not equivalent to, or considered prescription medications, and have variable fatty acid content
- Give with a fat-containing meal to ensure absorption (pancreatic lipase stimulation breaks ethyl bonds to facilitate absorption)
Common Side Effects:
Eructation
Dyspepsia
Taste perversion
Contraindications‖:
- History of hypersensitivity to the omega-3 fatty acid product
Comments:
- Side effects less with prescription omega-3 fatty acids compared to nonprescription fish oil supplements
- New onset atrial fibrillation/flutter reported
- Must monitor hepatic transaminases in patients with hepatic impairment
- DHA-containing products (omega-3 acid ethyl esters) can raise LDL-C, but not apoB
- Derived from fish and purified; use with caution in patients with known hypersensitivity to fish and/or shellfish
- Possible increased bleeding when used in combination with an antithrombotic agent
FDA-approved indication(s)§:
Icosapent ethyl (IPE):
VASCEPA is an ethyl ester of eicosapentaenoic acid (EPA) indicated:
- as an adjunct to maximally tolerated statin therapy to reduce the risk of myocardial infarction, stroke, coronary revascularization, and unstable angina requiring hospitalization in adult patients with elevated triglyceride (TG) levels (≥ 150 mg/dL) and:
- established cardiovascular disease or
- diabetes mellitus and 2 or more additional risk factors for cardiovascular disease.
- as an adjunct to diet to reduce TG levels in adult patients with severe (≥ 500 mg/dL) hypertriglyceridemia.
Limitations of Use:
- The effect of VASCEPA on the risk for pancreatitis in patients with severe hypertriglyceridemia has not been determined.
§Currently the FDA website only has label information for Vascepa, so its FDA indications are presented here.
Omega-3-acid ethyl esters:
LOVAZA is a combination of ethyl esters of omega 3 fatty acids, principally eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), indicated as an adjunct to diet to reduce triglyceride (TG) levels in adult patients with severe (≥500 mg/dL) hypertriglyceridemia.
Limitations of Use:
- The effect of LOVAZA on the risk for pancreatitis has not been determined.
- The effect of LOVAZA on cardiovascular mortality and morbidity has not been determined.
§Currently the FDA website only has label information for Lovaza, so its FDA indications are presented here.
Class: Niacin
Mechanism of Action: Reduces esterification of hepatic TG, decreases release of free fatty acids from adipose tissue and increases activity of lipoprotein lipase to enhance removal of TG-rich lipoprotein fatty acids
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Extended release niacin | 500-2000 mg | Once daily |
| Immediate release niacin | 250-6000 mg | Twice to 3 times daily |
Guideline Comments†:
- Oral agents
- CVOTs show no reduction in cardiovascular events when added to statin therapy in individuals being treated for secondary prevention of ASCVD
- Last-line agent for severe hypertriglyceridemia due to relatively high risk of intolerable side effects; must initiate low dose to minimize side effects and slowly titrate up to an effective dose
- Expected TG reduction: sustained-release, 10%-30%; immediate-release 20%-50%‡
- Increases insulin resistance, high rates of skin flushing, and significant hepatotoxicity
Common Side Effects:
- Facial flushing
- Gastrointestinal symptoms (diarrhea, nausea, vomiting) are common
- Persistent hepatic transaminase and glucose elevations
Contraindications‖:
- Active liver disease (including unexplained persistent elevated hepatic transaminases)
- Active peptic ulcer disease
- Arterial bleeding
- Known hypersensitivity to product components
Comments:
- Monitoring liver enzymes before and during treatment is necessary
- May cause and/or exacerbate insulin resistance
- Infrequently used owing to poor tolerability and lack of proven cardiovascular benefits; tolerability improved with long-acting products and by optimizing administration (slow titration, administering with food, predosing with aspirin or an NSAID)
- Serious muscle toxicity possible with statin use
FDA-approved indication(s)§:
Niacin extended-release tablets:
NIASPAN contains extended-release niacin (nicotinic acid), and is indicated:
- To reduce elevated TC, LDL-C, Apo B and TG, and to increase HDL-C in patients with primary hyperlipidemia and mixed dyslipidemia.
- To reduce the risk of recurrent nonfatal myocardial infarction in patients with a history of myocardial infarction and hyperlipidemia.
- In combination with a bile acid binding resin:
- Slows progression or promotes regression of atherosclerotic disease in patients with a history of coronary artery disease (CAD) and hyperlipidemia.
- As an adjunct to diet to reduce elevated TC and LDL-C in adult patients with primary hyperlipidemia.
- To reduce TG in adult patients with severe hypertriglyceridemia.
Limitations of use:
Addition of NIASPAN did not reduce cardiovascular morbidity or mortality among patients treated with simvastatin in a large, randomized controlled trial.
§Currently the FDA website only has label information for Niaspan, so its FDA indications are presented here.
Niacin immediate-release tablets:
FDA label for Niacor is available on FDA website.
Class: ApoC-III inhibitor: ASO directed therapy
Mechanism of Action: Binds to and degrades apoC-III mRNA; reduces the apoC-III protein, leading to increased clearance of plasma TG and VLDL
Medications, Typical Dose Range, and Dosing Frequency:
| Medications | Typical Dose Range | Dosing Frequency |
|---|---|---|
| Olezarsen | 80 mg | Once monthly |
Guideline Comments†:
- Subcutaneous agent
- Only approved for familial chylomicronemia syndrome
- Expected TG reduction in familial chylomicronemia syndrome: 30% (placebo-corrected is 42.5%)‡
Common Side Effects:
- Injection site reactions
Contraindications‖:
- Prior serious hypersensitivity to olezarsen or any of the excipients
Comments:
- Use is restricted to patients with FCS
- Decreased platelet count is possible but not common
FDA-approved indication(s)§:
Olezarsen:
TRYNGOLZA is an APOC-III-directed antisense oligonucleotide (ASO) indicated as an adjunct to diet to reduce triglycerides in adults with familial chylomicronemia syndrome (FCS).
§Currently the FDA website only has label information for Tryngolza, so its FDA indications are presented here.
*Dosages and administration from FDA-approved labeling.
†Adverse effects are discussed in Section 5.1, "Medication Safety and Therapy-Associated Side Effects."
‡Expected lipid-lowering based on estimations from the "2022 ACC Expert Consensus Decision Pathway on the Role of Nonstatin Therapies for LDL-Cholesterol Lowering in the Management of Atherosclerotic Cardiovascular Disease Risk," the "2019 AHA Science Advisory," or product labeling.
‖Contraindications from FDA-approved labeling (available at http://dailymed.nlm.nih.gov/dailymed/index.cfm)
ANGPTL3 indicates angiopoietin-like protein 3; ApoC-III, apolipoprotein C-III; ASO, antisense oligonucleotide; ATP, adenosine triphosphate; CK, creatine kinase; CVOT, cardiovascular outcome trial; DHA, docosahexaenoic acid; EPA, eicosapentaenoic acid; FCS, familial chylomicronemia syndrome; FH, familial hypercholesterolemia; HMG-CoA, 3-hydroxy-3-methylglutaryl- coenzyme; HoFH, homozygous familial hyperlipidemia; mRNA, messenger ribonucleic acid; NPC1L1, Niemann-Pick C1-Like1; NSAID, nonsteroidal anti-inflammatory drug; PCSK9, proprotein convertase subtilisin/kexin type 9; PPAR-alpha, peroxisome proliferator-activated receptor-alpha; REMS, Risk Evaluation and Mitigation Strategy; RNA, ribonucleic acid; TG, triglycerides; ULN, upper limit of normal; VLDL, very-low density lipoprotein; and XL, extended release.
Source: 2026 Guideline on the Management of Dyslipidemia, Tables 5, 25, and 26, with additional FDA indication information
Lipid-Lowering Therapies During Pregnancy and Lactation
| Pregnancy | Lactation | |
|---|---|---|
| Statins |
|
|
| Ezetimibe |
|
|
| Bile acid sequestrants |
|
|
| Niacin |
|
|
| Fibric acid derivatives |
|
|
| Omega-3 fatty acids |
|
|
| Bempedoic acid |
|
|
| PCSK9 mAb |
|
|
| Inclisiran |
|
|
| Evinacumab |
|
|
| Lomitapide |
|
|
Adapted with permission from Agarwala et al. © 2024 Elsevier. Adapted with permission from Jacobson et al. © 2015 Elsevier.
ASCVD indicates atherosclerotic cardiovascular disease; FH, familial hypercholesterolemia; mAb, monoclonal antibodies; and PCSK9, proprotein convertase subtilisin/kexin type 9.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 20
Considerations for Referral to a Lipid Specialist*
- Patients with diagnosed or suspected FH
- Patients with homozygous FH
- Patients with heterozygous FH who do not achieve treatment targets on maximally tolerated statin plus nonstatin therapy
- Patients with heterozygous FH with statin-attributed side effects on ≥2 statins, including at the lowest dose or with alternate dosing regimens
- Patients with ASCVD or at high risk of ASCVD
- Patients with premature ASCVD (onset age <40 years)
- Patients who do not achieve ≥50% LDL-C reduction and LDL-C (or non–HDL-C) targets on maximally tolerated statin plus nonstatin therapy
- Patients with statin-attributed side effects on ≥2 statins, including at the lowest dose or with alternate dosing regimens
- Patients who have elevated Lp(a) (≥200 nmol/L or ≥75 mg/dL)
- Patients <40 years old with diabetes and dyslipidemia
- Patients at high risk for ASCVD or with ASCVD who are on complex medication regimens
- Patients receiving treatment for HIV
- Patients receiving treatment for cancer
- Patients receiving treatments to prevent transplant rejection
- Individuals who are considering pregnancy, are pregnant, or are breastfeeding
- Patients with heterozygous FH
- Patients with hypertriglyceridemia (TG ≥400 mg/dL)
- Patients with ASCVD or at high risk of ASCVD requiring LLT
- Patients with inherited hyperlipidemias who need genetic testing for diagnosis
- Patients with severe/extreme primary hypertriglyceridemia after secondary causes have been ruled out
- Patients who may be candidates for treatment with evinacumab, lomitapide, olezarsen, or lipoprotein apheresis
*Especially if patients are not achieving lipid/lipoprotein goals on recommended therapies.
ASCVD indicates atherosclerotic cardiovascular disease; FH, familial hypercholesterolemia; HDL-C, high-density lipoprotein-cholesterol; HIV, human immunodeficiency virus; LDL-C, low-density lipoprotein-cholesterol; LLT, lipid-lowering therapy; LP(a), lipoprotein (a); and TG; triglycerides.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 9
Predicting Risk of Cardiovascular Disease EVENTs equations (PREVENT™)
The PREVENT™-ASCVD equations utilized within this app are part of the broader PREVENT™ risk model developed by the American Heart Association which estimates 10- and 30-year risk for total CVD, ASCVD, and heart failure (HF). This risk model incorporates kidney and metabolic health measures such as BMI and estimated glomerular filtration rate (eGFR), and supporting optional predictors including urine albumin creatinine ratio (UACR), hemoglobin A1c (HbA1c), and social deprivation index (SDI). The complete PREVENT™ risk model as well as the 2013 ACC/AHA Pooled Cohort Equations (previously known as the ASCVD Risk Estimator) can be found in ACC’s CVD Risk Estimator Plus.
The PREVENT™ equations were developed by select members of the American Heart Association Cardiovascular-Kidney-Metabolic Scientific Advisory Group. The risk equations were derived and validated in a large, diverse sample of over 6 million individuals.
- Khan SS, Matsushita K, Sang Y, et al. Development and Validation of the American Heart Association Predicting Risk of Cardiovascular Disease EVENTs (PREVENTTM) Equations. Circulation 2023. DOI: 10.1161/CIRCULATIONAHA.123.067626.
- Khan SS, Coresh J, Pencina MJ, et al. Novel Prediction Equations for Absolute Risk Assessment of Total Cardiovascular Disease Incorporating Cardiovascular-Kidney-Metabolic Health: A Scientific Statement From the American Heart Association. Circulation 2023;148(24):1982-2004. DOI: 10.1161/CIR.0000000000001191.
The information derived from the use of PREVENT™ is based on PREVENT™ 1.0.0. Updates and future adaptations of PREVENT™ may yield different results and conclusions.
The American College of Cardiology Foundation is an independent entity and is not affiliated with, endorsed by, sponsored by, or approved by American Heart Association (“AHA”). Use of the PREVENT™ risk model and related materials is pursuant to a license from AHA and does not imply any partnership or association.
Groups that Benefit from Lipid-Lowering Therapies
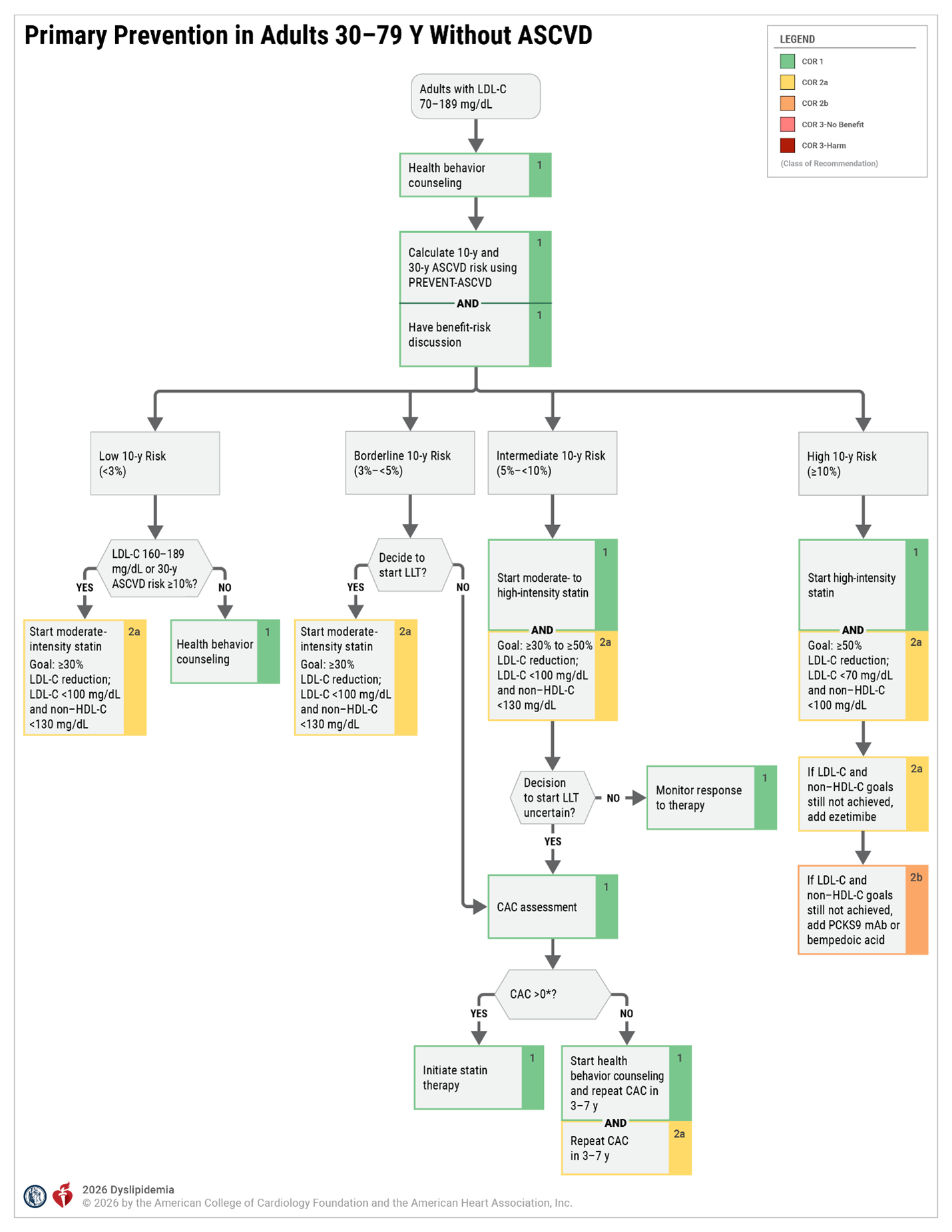
*For primary prevention recommendations for risk assessment and/or management in the following patient groups, please see the following: Section 4.2.3.7, "Primary Prevention in Adults 30 to 79 Years of Age With LDL-C Levels 70 to 189 mg/dL (1.8-4.9 mmol/L)," Section 4.2.4, "Severe Hypercholesterolemia (LDL-C ≥190 mg/dL [4.9 mmol/L])," Section 4.2.5, “Diabetes in Adults Without Established ASCVD," Section 4.2.7, "Management of Adults With Subclinical Coronary Atherosclerosis (Men ≥40 or Women ≥45 Years),” Section 4.2.8.8, "Adults With CKD—Stage 3 or Higher", and Section 4.3.8.9, “Persons Living With HIV."
ASCVD indicates atherosclerotic cardiovascular disease; CAC, coronary artery calcium; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; and LLT, lipid-lowering therapy. Adapted with permission from Grundy at al. © Copyright 2018 American Heart Association, Inc. and American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 6
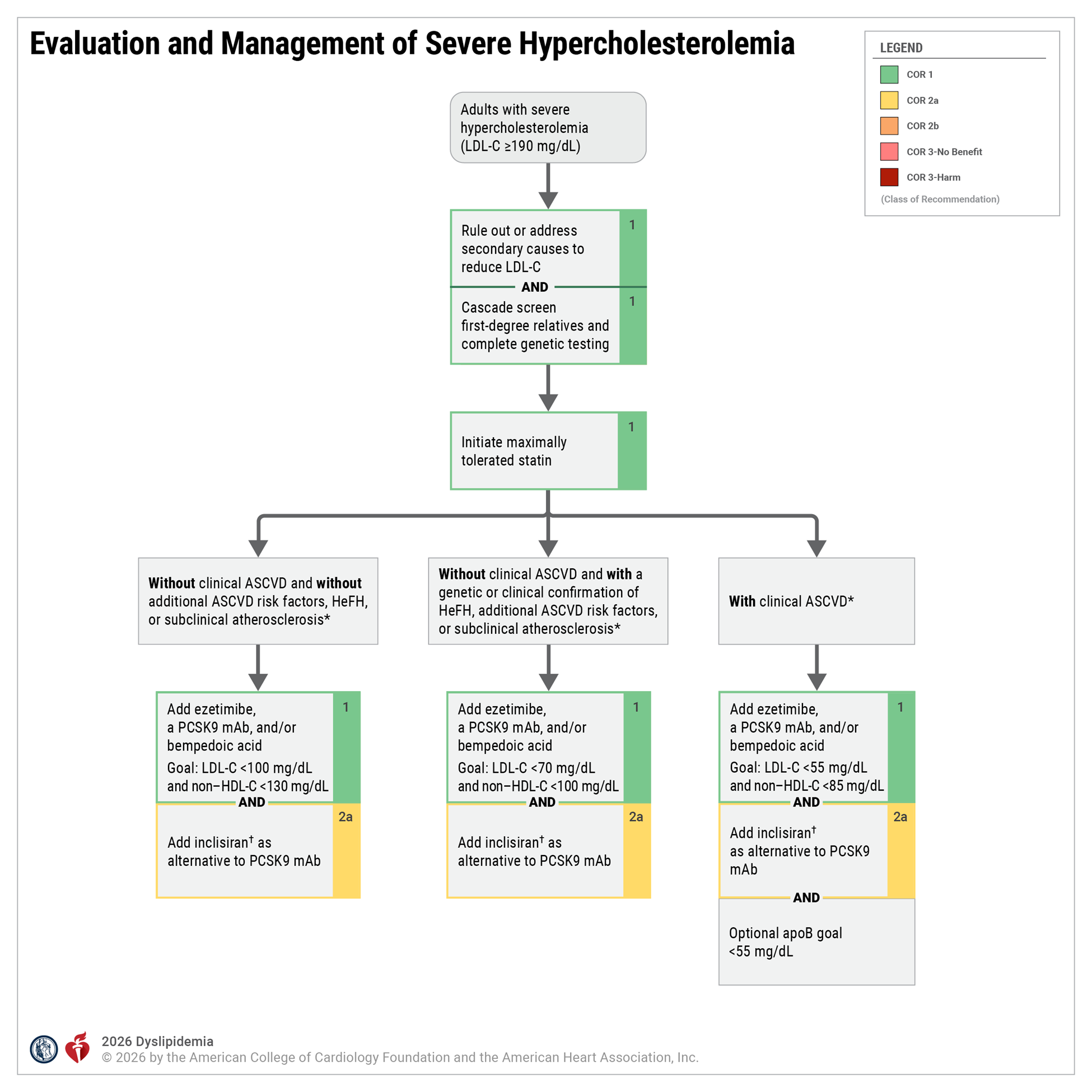
*Related to Lipoprotein Goals for ASCVD Risk Reduction table. †Cardiovascular outcomes trials with inclisiran are pending. ApoB indicates apolipoprotein B; ASCVD, atherosclerotic cardiovascular disease; CAC, coronary artery calcium; HDL-C, high-density lipoprotein cholesterol; HeFH, heterozygous familial hypercholesterolemia; LDL-C, low-density lipoprotein cholesterol; LLT, lipid-lowering therapy; mAb, monoclonal antibody; and PCSK9, proprotein convertase subtilisin/kexin type 9.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 7
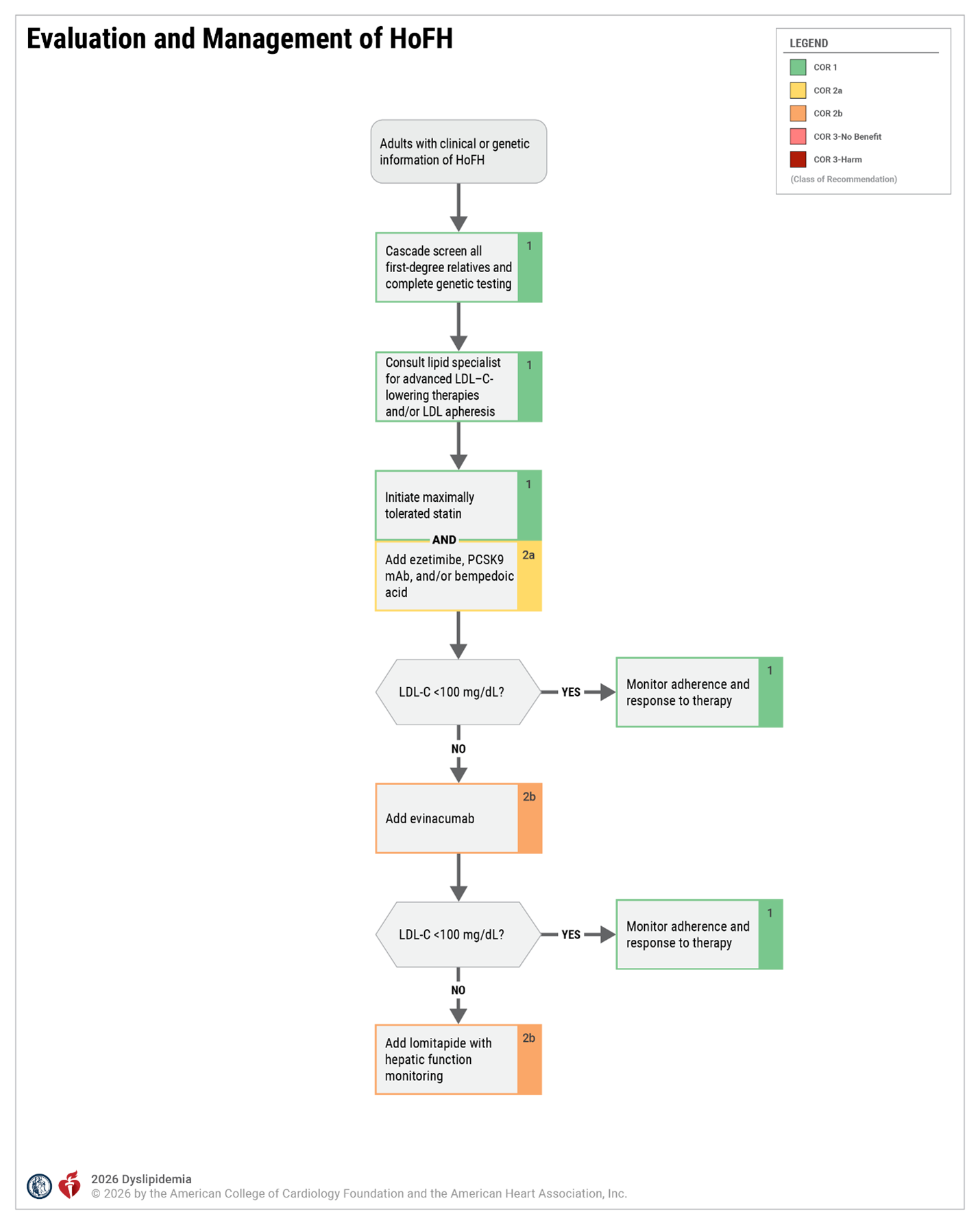
HoFH indicates homozygous familial hypercholesterolemia; LDL-C, low-density lipoprotein cholesterol; and PCSK9, proprotein convertase subtilisin/kexin type 9.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 8
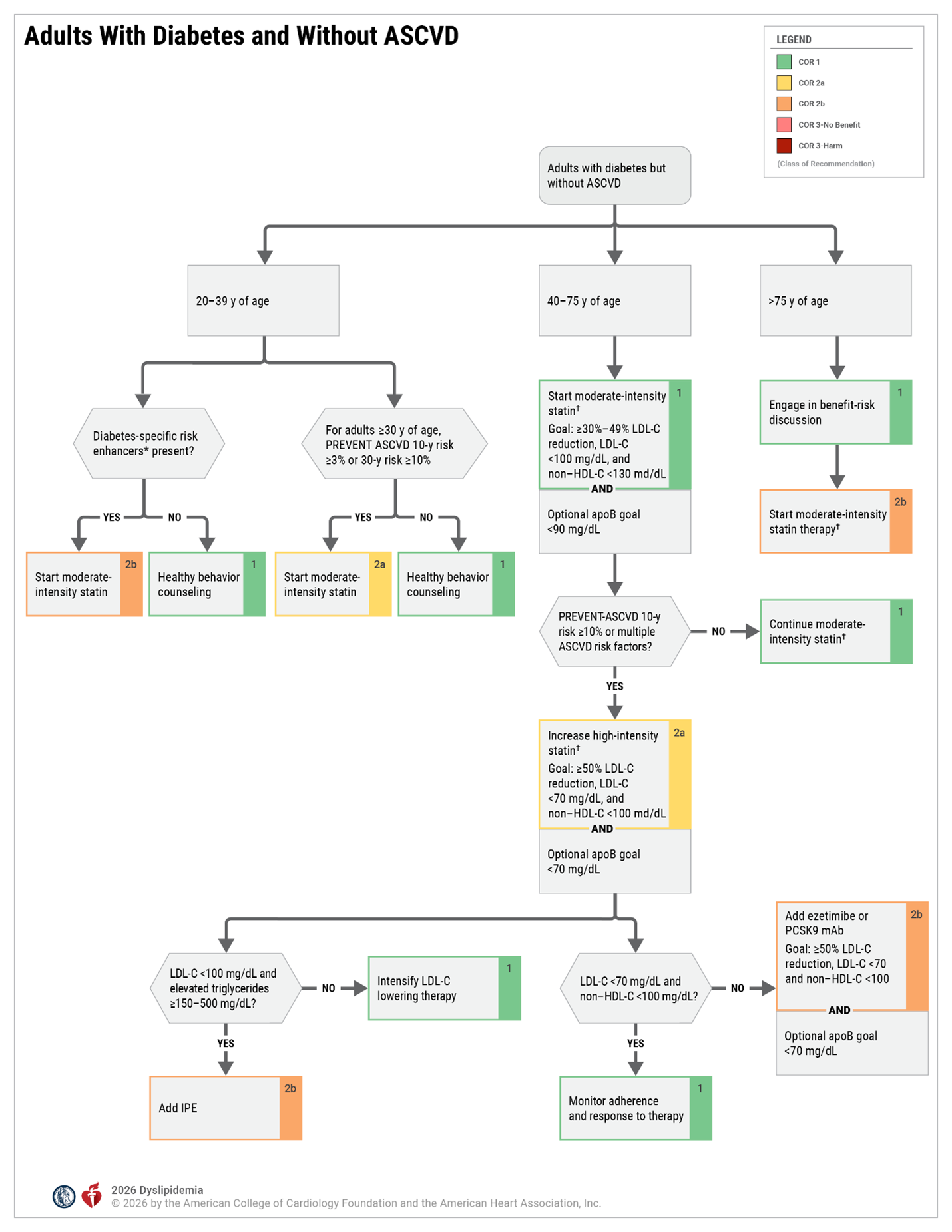
apoB indicates apolipoprotein B; ASCVD, atherosclerotic cardiovascular disease; HDL-C, high-density lipoprotein cholesterol; IPE, icosapent ethyl; LDL-C, low-density lipoprotein cholesterol; LLT, lipid-lowering therapy; mAb, monoclonal antibody; and PCSK9, proprotein convertase subtilisin/kexin type 9.
*Refer to Table 17, "Diabetes-Specific Risk Enhancers Independent of Other Diabetes-Related Risk Factors."
†In adults with diabetes who have statin-attributed side effects, initiation of ezetimibe and/or bempedoic acid and/or a PCSK9 mAb is recommended to lower LDL-C and reduce ASCVD risk.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 9
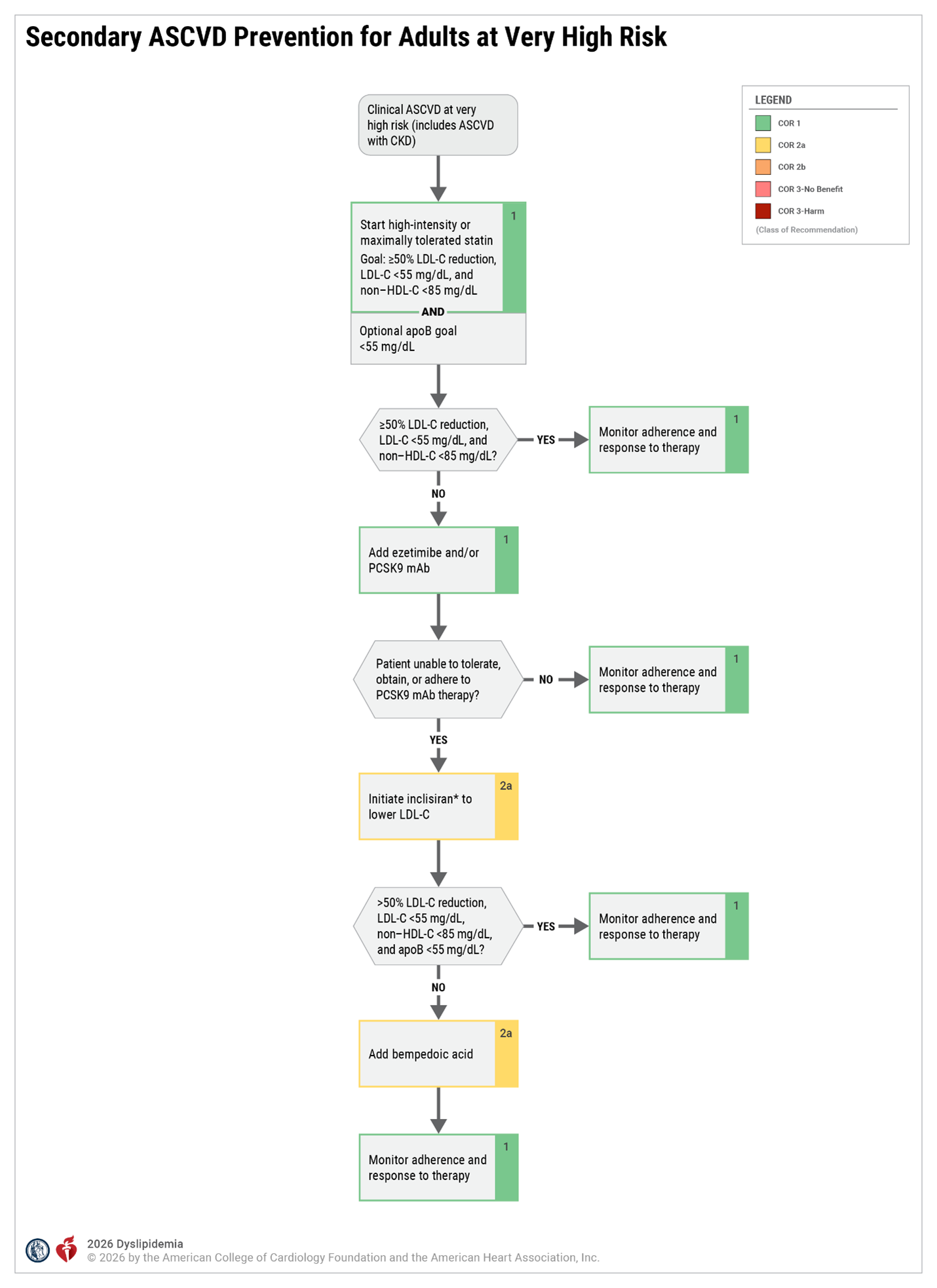
*Two cardiovascular outcome studies in patients with ASCVD are ongoing with inclisiran. ASCVD indicates atherosclerotic cardiovascular disease; CKD, chronic kidney disease; HDL-C, high-density lipoprotein cholesterol; HeFH, heterozygous familial hypercholesterolemia; LDL-C, low-density lipoprotein cholesterol; mAb, monoclonal antibody; and PCSK9, proprotein convertase subtilisin/kexin type 9.
Adapted with permission from Grundy at al. © 2018 American Heart Association, Inc. and American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 11
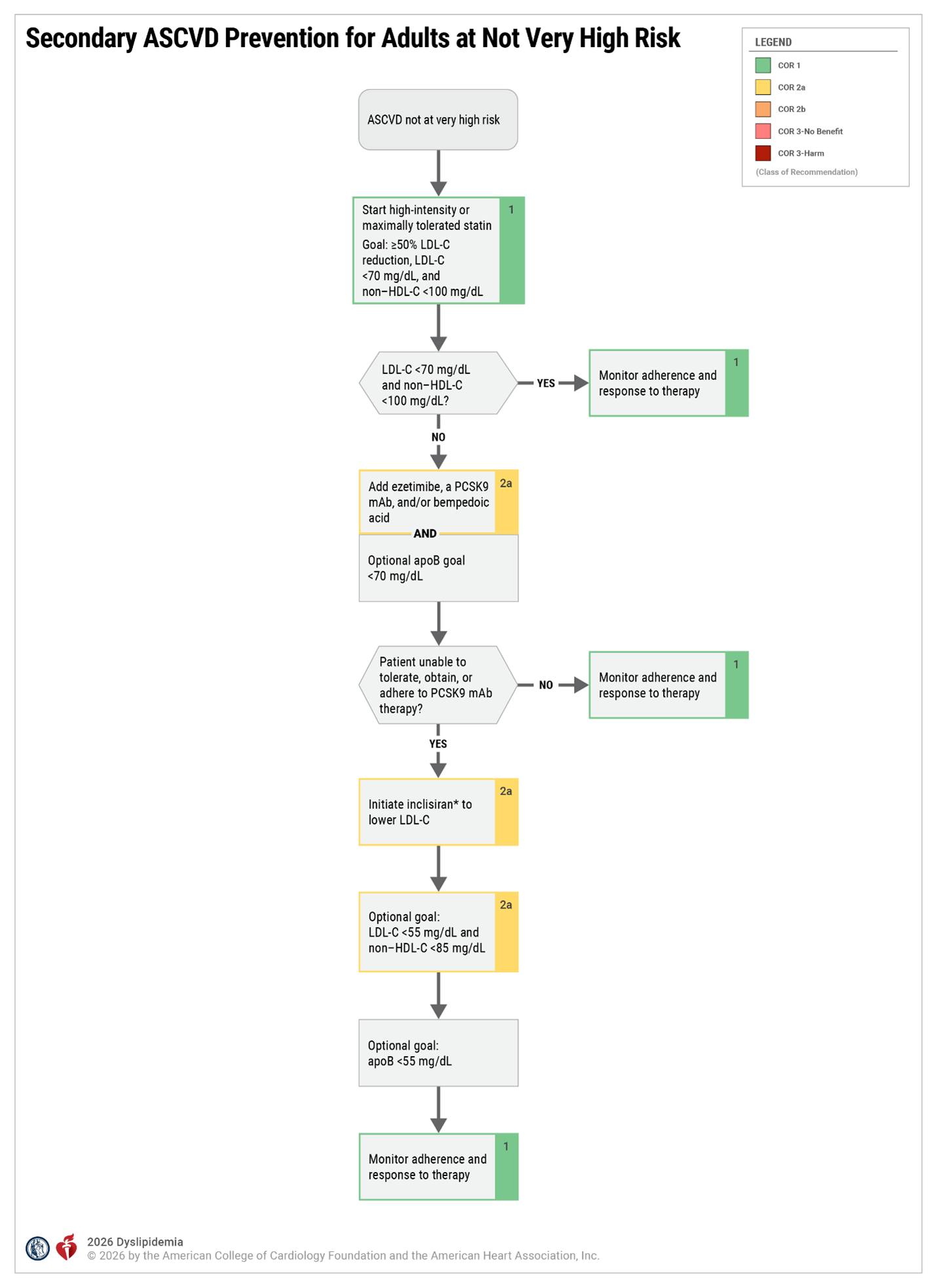
*CVOTS pending; the majority of patients with ASCVD are at very high risk. ASCVD indicates atherosclerotic cardiovascular disease; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; mAb, monoclonal antibody; and PCSK9, proprotein convertase subtilisin/kexin type 9.
Adapted with permission from Grundy at al. © 2018 American Heart Association, Inc. and American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 12
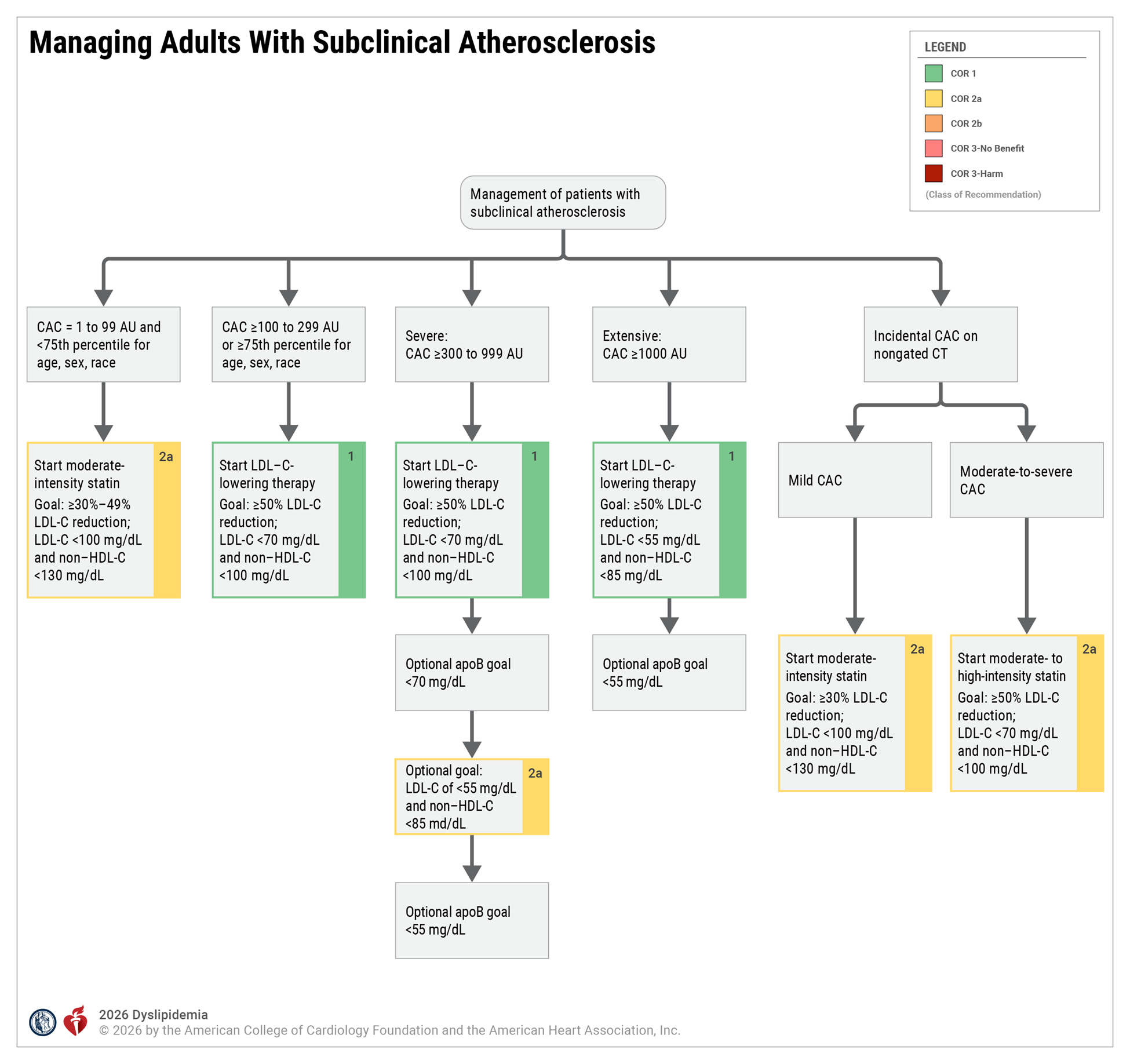
apoB indicates apolipoprotein B; AU, Agatston Units; CAC, coronary artery calcium; CT, computed tomography; HDL-C, high-density lipoprotein cholesterol; and LDL-C, low-density lipoprotein cholesterol.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 13
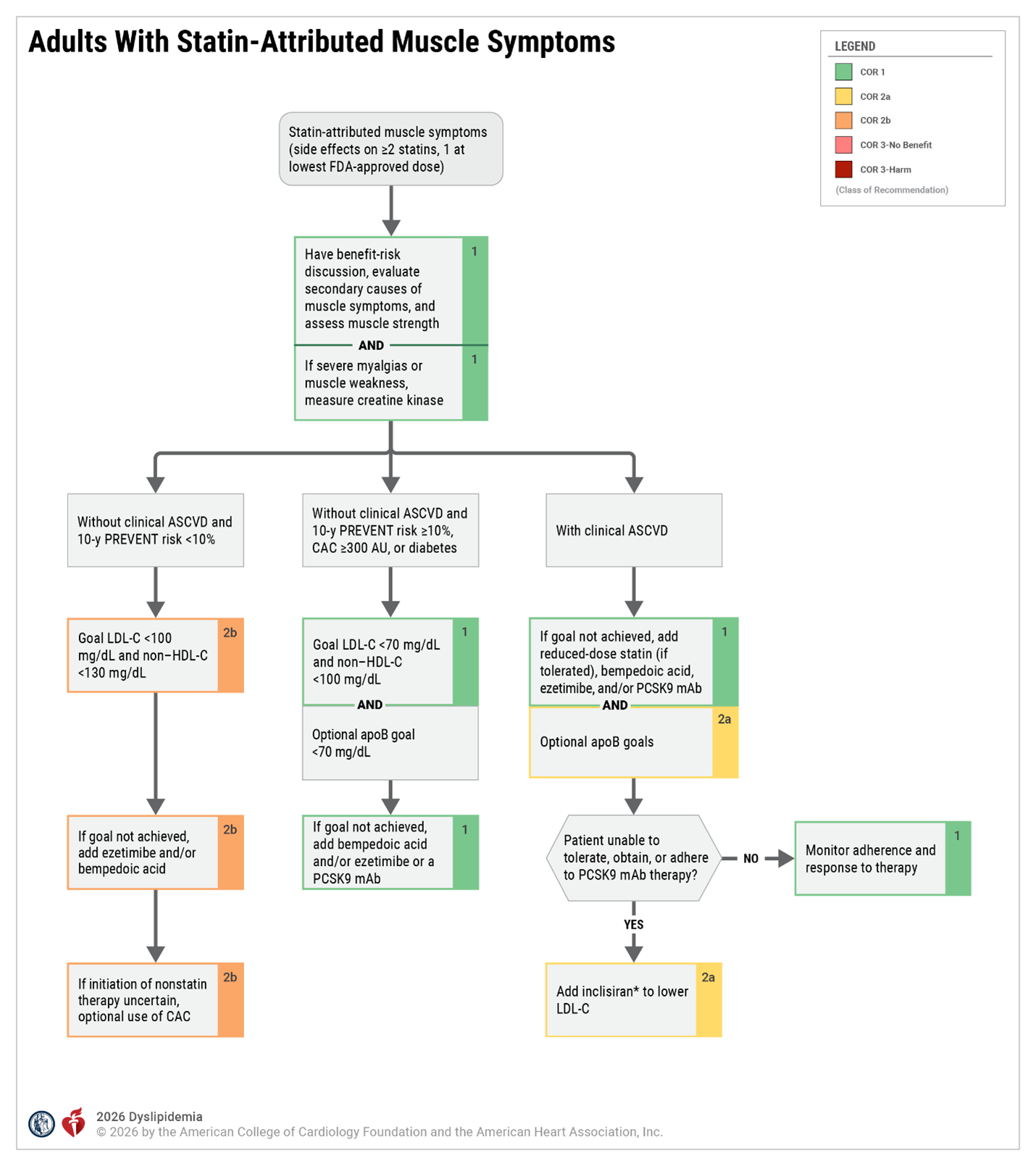
*Cardiovascular outcomes for inclisiran pending results of ongoing RCTs. ASCVD indicates atherosclerotic cardiovascular disease; CAC, coronary artery calcium; FDA, U.S. Food and Drug Administration; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; LLT, lipid-lowering therapy; mAb, monoclonal antibody; and PCSK9, proprotein convertase subtilisin/kexin type 9.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 18
Clinical ASCVD: Criteria for Defining "At Very High Risk" in Adults

ACS indicates acute coronary syndrome; ASCVD, atherosclerotic cardiovascular disease; Hx, history; LDL-C, low-density lipoprotein cholesterol; MI, myocardial infarction; and PAD, peripheral artery disease.
Adapted with permission from Grundy et al. © 2018 American Heart Association, Inc. and American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 10
Risk-Enhancing Factors for Clinician-Patient Risk Discussion
Risk Enhancers
- History of premature ASCVD in a parent or sibling (onset age <55 y for men, <65 y for women)
- Higher risk ancestry (eg, South Asian, Filipino)
- High polygenic risk (if measured) (Section 4.2.3.5, “Polygenic Risk Scores”)
- Chronic inflammatory diseases (eg, systemic lupus, rheumatoid arthritis, advanced psoriasis, inflammatory arthritis)
- Lp(a) ≥125 nmol/L or ≥50 mg/dL
- hsCRP ≥2 mg/L on >1 occasion (if measured)
- TG persistently ≥175 mg/dL (2 mmol/L) (if nonfasting) and ≥150 mg/dL (1.7 mmol/L) (if fasting)
- CKM syndrome
- LDL-C persistently ≥160-189 mg/dL (4.1-4.9 mmol/L), non–HDL-C ≥190-219 mg/dL or apoB ≥120 mg/dL*
- Reproductive risk markers (premature menopause, preeclampsia, gestational diabetes, gestational hypertension, preterm delivery; Section 4.2.3.4, “Reproductive Risk Marker”)
Note that all available information should be included in risk estimates derived from the PREVENT-ASCVD equations, including albuminuria, HbA1c, and zip code for assessment of neighborhood-level social determinants of health. Given the recent publication of the PREVENT-ASCVD equations, it remains to be demonstrated for most risk enhancers that risk is incremental to the PREVENT-ASCVD equations.
*Although LDL-C is not included in the PREVENT-ASCVD equations (total cholesterol and HDL-C are included), it is included here because persistent elevation of LDL-C may be a useful factor to include in risk-benefit discussions about LDL-C–lowering therapy, given that it is the target of that therapy. See Section 4.2.3.4, "Reproductive Risk Marker," for more detail regarding reproductive risk factors.
ApoB indicates apolipoprotein B; ASCVD, atherosclerotic cardiovascular disease; CKM, cardiovascular-kidney-metabolic; HbA1c, hemoglobin A1c; HDL-C, high-density lipoprotein-cholesterol; hsCRP, high-sensitivity C-reactive protein; LDL-C, low-density lipoprotein-cholesterol; Lp(a), lipoprotein (a); and TG, triglycerides.
Adapted with permission from Grundy et al. © Copyright 2018 American Heart Association, Inc. and American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 13
Reproductive Risk Markers Associated With ASCVD Events
Adverse Pregnancy Outcomes* That Have a Stronger Association With ASCVD Events
- Hypertensive disorders of pregnancy (preeclampsia, gestational hypertension)
- Gestational diabetes
- Small-for-gestational age (birthweight below the 10th percentile)
- Preterm delivery (before 37 wk gestation)
- Recurrent spontaneous pregnancy loss
Other Reproductive Risk Markers
- Early menarche (<10 y old)
- Early menopause (<45 y old), especially premature menopause (<40 y old)
- Polycystic ovarian syndrome and irregular menses
*See reference for a full list of Adverse Pregnancy Outcomes.
ASCVD indicates atherosclerotic cardiovascular disease.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 14
Physiological and Secondary Causes of Hypercholesterolemia Due to LDL-C
Dietary factors
- High saturated fat intake
- High trans-fat intake
- High cholesterol intake
- Weight gain
- Rapid weight loss
- Ketosis
Metabolic factors
- Hypothyroidism
- Obstructive liver disease
- Chronic kidney disease
- Nephrotic syndrome
- Diabetes and other insulin-resistant states (excess small LDL particles)
- Uncontrolled hyperglycemia
- Cushing syndrome
- Anorexia nervosa
- Obesity
Drugs
- High-dose thiazide diuretics
- Glucocorticoids
- Estrogens
- Androgens
- Atypical antipsychotic drugs
- Cyclosporine
Physiological
- Menopausal transition
- Pregnancy
LDL indicates low-density lipoprotein. Modified with permission from Virani et al.
© 2021 American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 15
Diabetes-Specific Risk Enhancers Independent of Other Diabetes-Related Risk Factors
Risk Enhancers
- Long duration (≥10 y for type 2 diabetes or ≥20 y for type 1 diabetes)
- Albuminuria ≥30 μg of albumin/mg creatinine
- eGFR <60 mL/min/1.73 m²
- Retinopathy
- Neuropathy
- ABI <0.9
Reprinted with permission from Grundy et al. © 2018 American Heart Association, Inc. and American College of Cardiology Foundation.
ABI indicates ankle-brachial index; and eGFR, estimated glomerular filtration rate.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 17
Checklist for Individualized Benefit-Risk Discussion
| Checklist Item | Recommendation |
|---|---|
| ✓ ASCVD risk assessment |
|
| ✓ Emphasize healthy lifestyle habits as the foundation of treatment |
|
| ✓ Potential net clinical benefit of pharmacotherapy |
|
| ✓ Cost and convenience considerations |
|
| ✓ Shared decision-making |
|
AHA indicates American Heart Association; ASCVD, atherosclerotic cardiovascular disease; CAC, coronary artery calcium; CT, computed tomography; ECG, electrocardiogram; LDL-C, low-density lipoprotein-cholesterol; LLT, lipid-lowering therapy; PCNA, Preventive Cardiology Nurses Association; and NLA, National Lipid Association.
Adapted with permission from Martin et al. © Copyright 2015 American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 10
Physiological and Secondary Causes of Hypertriglyceridemia
| Categories | Conditions and Medications Contributing to Hypertriglyceridemia |
|---|---|
| Diseases |
|
| Diet/Lifestyle |
|
| Drugs* (Medications) |
Anesthesia:
|
| Disorders of metabolism |
|
| Physiological |
|
*Caveats: TG-raising medications require careful monitoring; minimizing other conditions that raise TG; and, when clinically appropriate, using alternatives.
TG indicates triglycerides. Adapted from Virani et al.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 23
Health Behavior Interventions in Patients With Hypertriglyceridemia

*Referral to an RDN and lipid specialist advised.
†Referral to an RDN necessary.
‡Clinicians may opt to reduce total fat as a percent of calories in some patients to 10% to 15% (examples include those with a history of pancreatitis or those at the higher end of this range).
§Limitation of total fat to 10% to 15% of total daily intake, with guidance from an RDN is essential for patients with FCS.
¶For those with TG disorders outside of FCS, individually tailored total fat limitation under the guidance of an RDN can be beneficial due to variable response to diet.
||Although clinicians should aim for a patient to achieve the guideline-directed amount of physical activity, any physical activity is likely beneficial in sedentary individuals and should be encouraged to reduce cardiometabolic risk.
FCS indicates familial chylomicronemia syndrome; RDN, registered dietician nutritionist; and TG, triglycerides.
Adapted with permission from Virani et al, with additional information obtained from Arnett et al. © 2021 American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 2
Groups that Benefit from Management of Hypertriglyceridemia
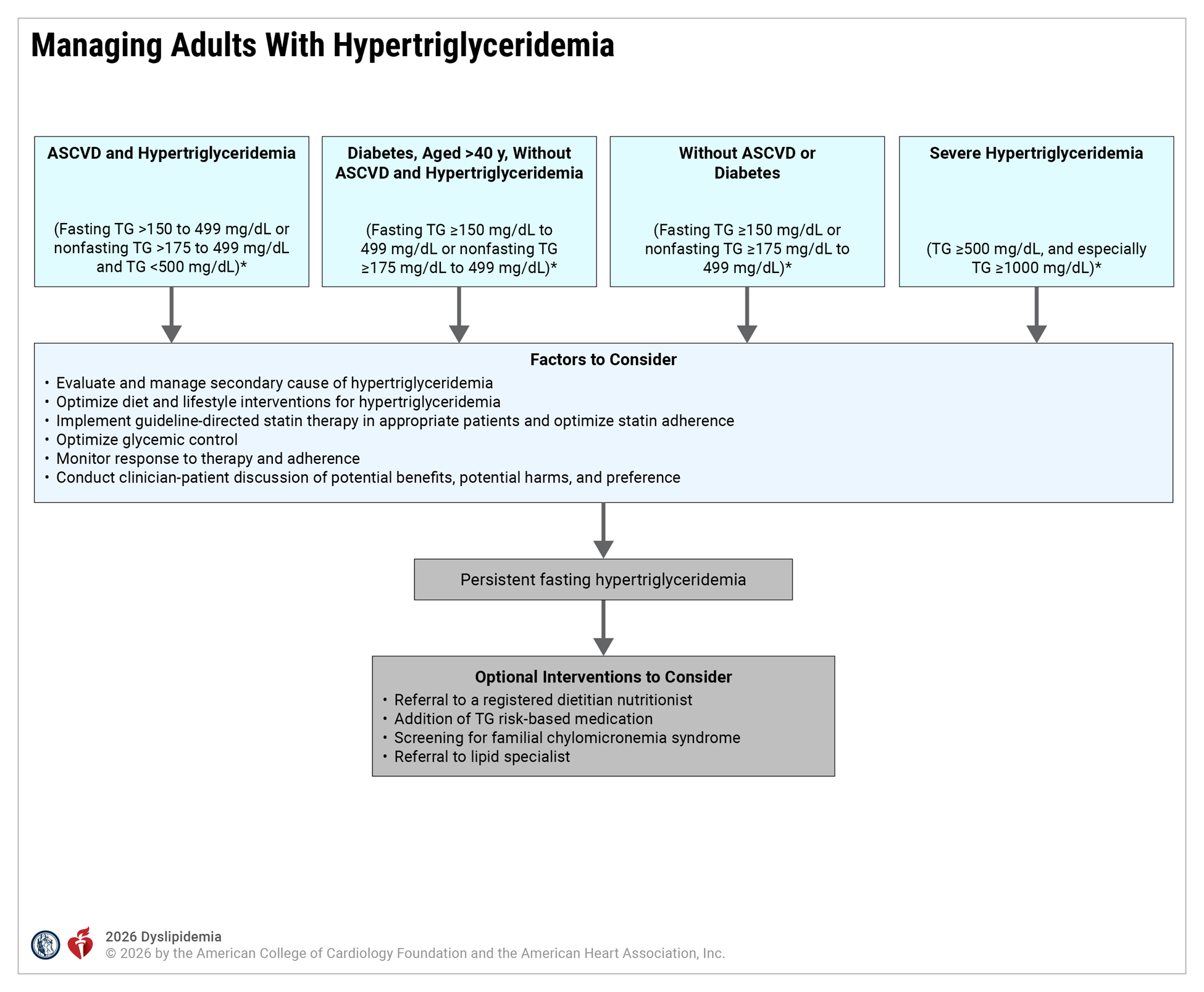
*Conversion of TG from mg/dL to mmol/L: 150 mg/dL=1.7 mmol/L, 175 mg/dL=2 mmol/L, 500 mg/dL to 5.7 mmol/L, 1000 mg/dL=11.3 mmol/L.
ASCVD indicates atherosclerotic cardiovascular disease; and TG, triglycerides. Adapted with permission from Virani et al. © 2021 American College of Cardiology Foundation.
Persistent hypertriglyceridemia is defined as fasting TG ≥150 mg/dL (1.7 mmol/L) after evaluation and management of secondary causes, a minimum of 4 to 12 weeks of lifestyle management, and on a stable, maximally tolerated dose of statin if indicated based on ASCVD risk.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 14
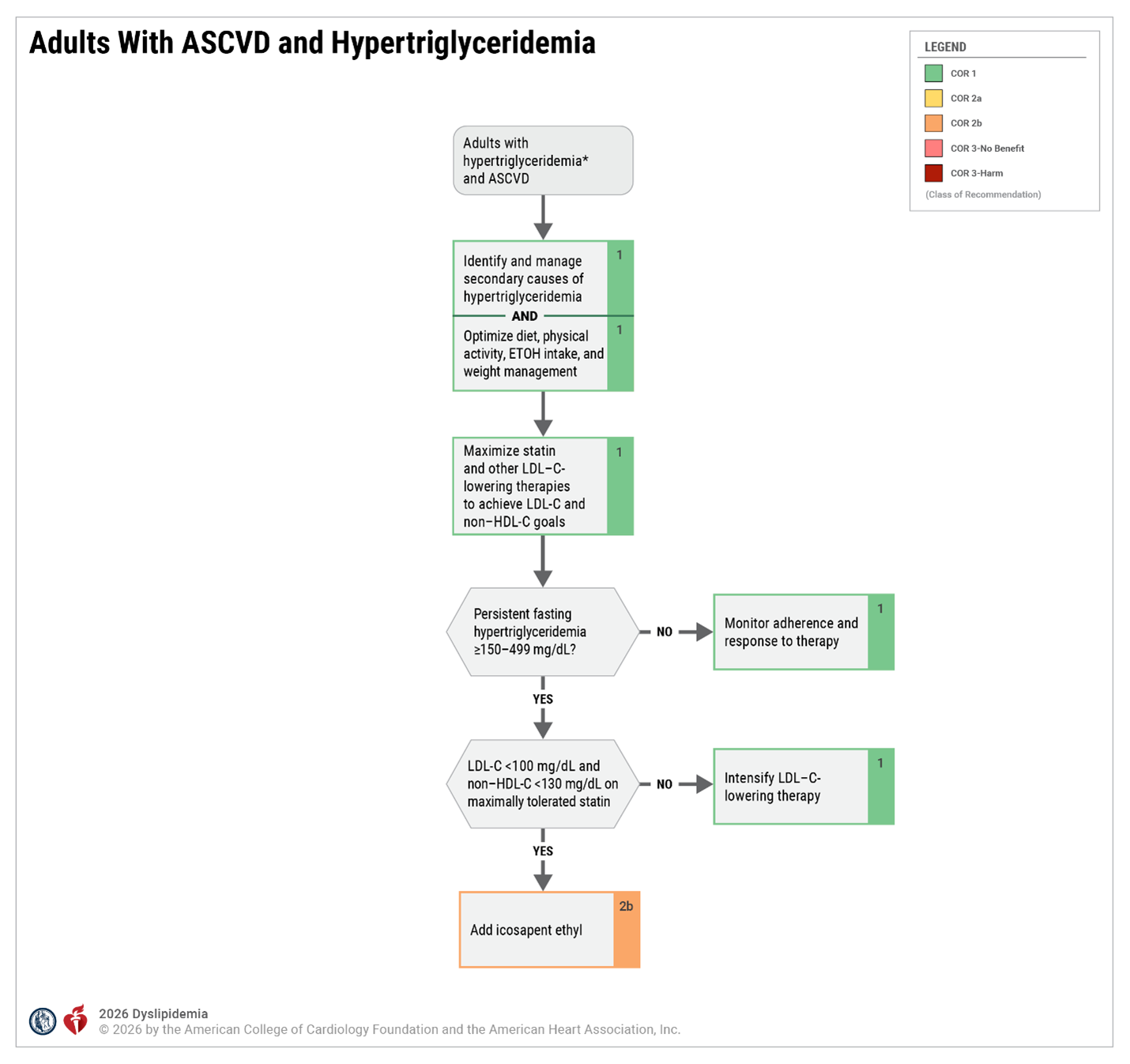
*Conversion of triglyceride from mg/dL to mmol/L: 150 mg/dL=1.7 mmol/L, 175 mg/dL=2 mmol/L, 500 mg/dL to 5.7 mmol/L, 1000 mg/dL=11.3 mmol/L.
ASCVD indicates atherosclerotic cardiovascular disease; ETOH, ethyl alcohol; HDL-C, high-density lipoprotein cholesterol; and LDL-C, low-density lipoprotein cholesterol. Adapted from Virani et al.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 15
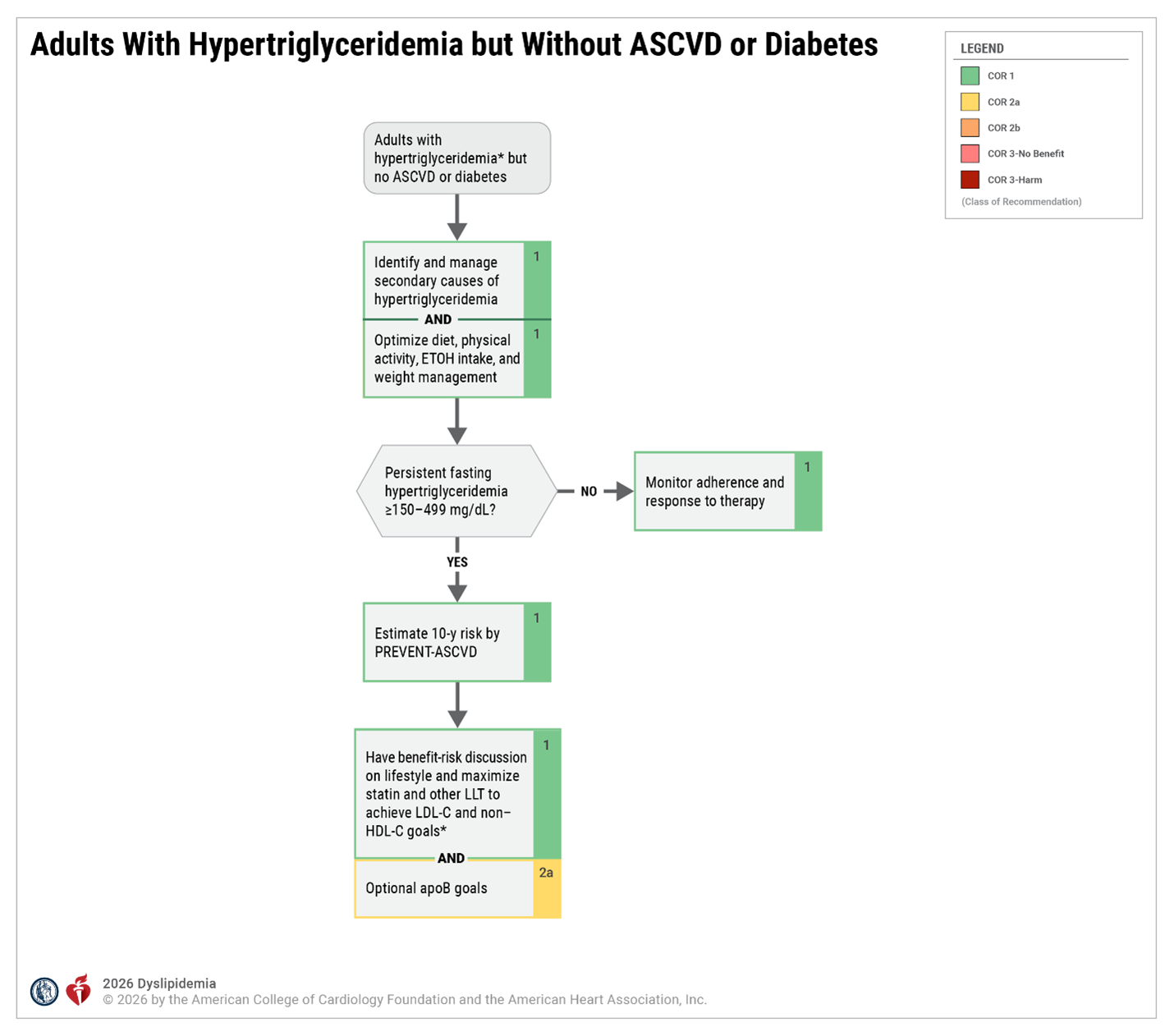
*Conversion of triglyceride from mg/dL to mmol/L: 150 mg/dL=1.7 mmol/L, 175 mg/dL=2 mmol/L, 500 mg/dL to 5.7 mmol/L, 1000 mg/dL=11.3 mmol/L.
apoB indicates apolipoprotein B; ASCVD, atherosclerotic cardiovascular disease; ETOH, ethyl alcohol; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; and LLT, lipid-lowering therapy. Adapted with permission from Virani et al. © 2021 American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 16
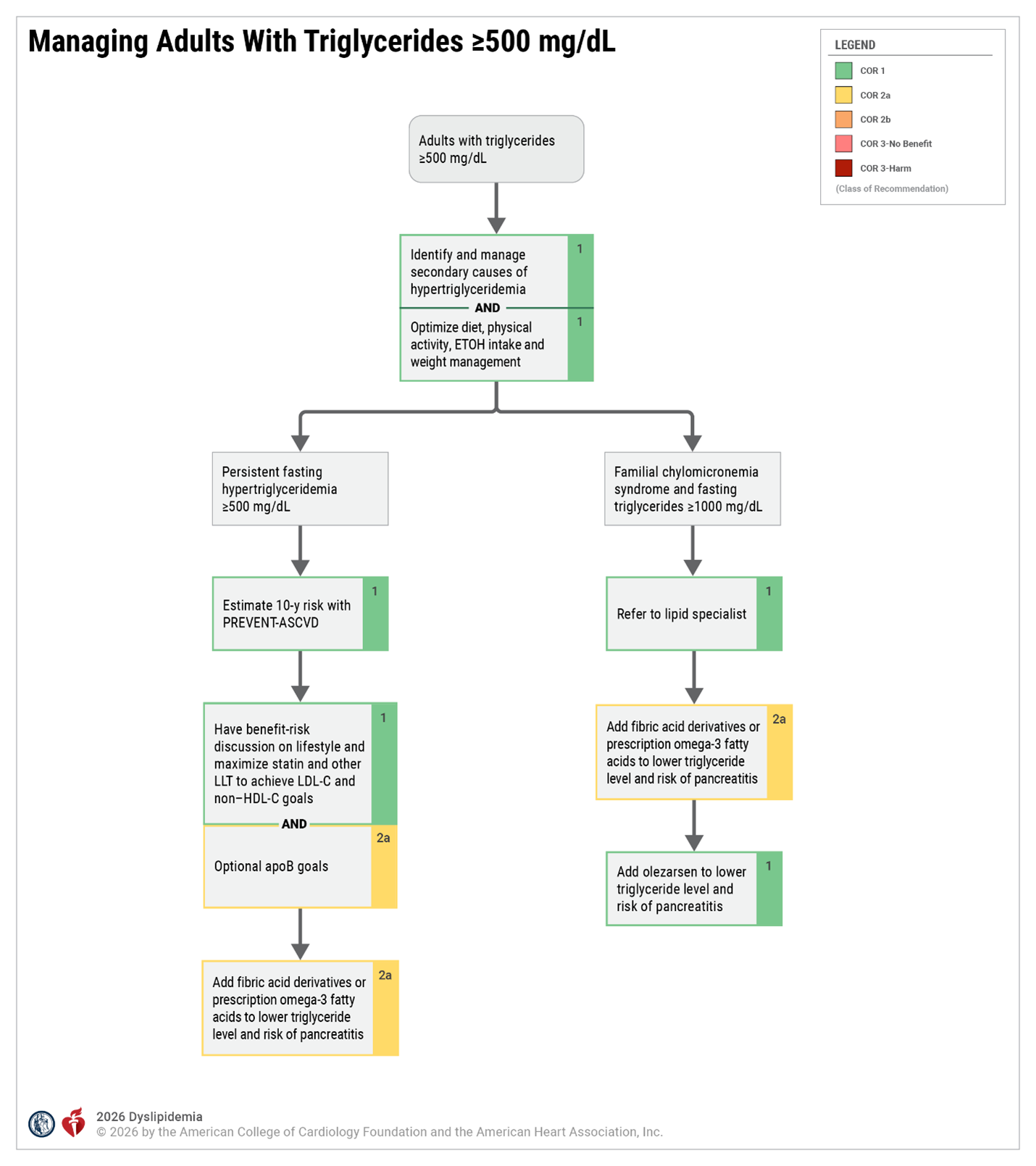
apoB indicates apolipoprotein B; ASCVD, atherosclerotic cardiovascular disease; ETOH, ethyl alcohol; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; and LLT, lipid-lowering therapy. Adapted with permission from Virani et al. © 2021 American College of Cardiology Foundation.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Figure 17
Patients or Characteristics Associated With Increased Risk for Statin-Attributed Muscle Symptoms
- Age ≥65 y
- Low body mass index
- Female sex
- Obesity
- Hypothyroidism
- Diabetes
- Chronic liver disease
- Chronic kidney disease
- Alcohol consumption
- Vigorous exercise
- High-dose statin therapy
- Diseases associated with myalgia or muscle weakness (eg, fibromyalgia, polymyalgia rheumatica, polymyositis, primary myopathies)
- Pharmacotherapy affecting statin metabolism (Section 5.2, “Statin-Cardiovascular Drug Interactions”)
- Gene variants affecting statin metabolism (eg, SLCO1B1)
An increased risk of statin-attributed muscle symptoms is associated with the use of higher doses of a statin within the approved dosage range. The increase in risk is not attributed to the statin potency (higher potency statins do not have higher risk; high doses do have higher risk). Adapted from Wiggins et al. Modified with permission from Stroes et al.
© 2015 Oxford University Press. Modified with permission via Creative Commons CC BY license from Bytyci et al. © 2022 Oxford University Press.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 24
Management of Statin Drug-Drug Interactions
Common Medications That May Interact With Statins
Avoid use with any statin
- Gemfibrozil
Can be used with a statin using a risk mitigation strategy*
- Amiodarone
- Amlodipine
- Atazanavir plus ritonavir
- Boceprevir
- Clarithromycin
- Cobicistat-containing products
- Colchicine
- Cyclosporine
- Danazol
- Darunavir plus ritonavir
- Diltiazem
- Dronedarone
- Erythromycin
- Fenofibrate
- Fenofibric acid
- Fluconazole
- Fosamprenavir (with or without ritonavir)
- Itraconazole
- Ketoconazole
- Lomitapide
- Lopinavir plus ritonavir
- Nefazodone
- Nelfinavir
- Niacin (≥1 g/d)
- Nirmatrelvir plus ritonavir
- Posaconazole
- Ranolazine
- Rifampin
- Saquinavir plus ritonavir
- Telaprevir
- Telithromycin
- Tipranavir plus ritonavir
- Verapamil
- Voriconazole
- Warfarin
*Risk mitigation strategies include a) avoiding use of the coadministered interacting medication; b) using an alternative statin that does not have the drug-drug interaction; and c) limiting the statin dose depending upon the statin and the nature of the drug-drug interaction. Modified with permission from Wiggins et al. © 2016 American Heart Association, Inc.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 8
Clinical Recommendations on Management of DDI With Statins and Cardiovascular Medications
| Interacting Drug | Statin | Clinical Recommendation for Management |
|---|---|---|
| Amiodarone | Lovastatin | Limit dose of lovastatin to 40 mg daily |
| Simvastatin | Limit dose of simvastatin to 20 mg daily | |
| Amlodipine | Lovastatin | Limit dose of lovastatin to 20 mg daily |
| Simvastatin | Limit dose of simvastatin to 20 mg daily | |
| Bempedoic acid | Pravastatin | Limit dose of pravastatin to 40 mg daily |
| Simvastatin | Limit dose of simvastatin to 20 mg daily | |
| Colchicine | Atorvastatin | Closer monitoring for muscle-related toxicity is recommended when used in combination |
| Fluvastatin | ||
| Lovastatin | ||
| Pitavastatin | ||
| Pravastatin | ||
| Rosuvastatin | ||
| Simvastatin | ||
| Conivaptan | Lovastatin | Avoid combination |
| Simvastatin | Avoid combination | |
| Diltiazem | Lovastatin | Limit dose of lovastatin to 20 mg daily |
| Simvastatin | Limit dose of simvastatin to 10 mg daily | |
| Dronedarone | Lovastatin | Limit dose of lovastatin to 10 mg daily |
| Simvastatin | Limit dose of simvastatin to 10 mg daily | |
| Gemfibrozil | Atorvastatin | Avoid combination |
| Lovastatin | ||
| Pitavastatin | ||
| Pravastatin | ||
| Rosuvastatin | ||
| Simvastatin | ||
| Lomitapide | Lovastatin | Reduce dose of lovastatin by 50% |
| Simvastatin | Reduce dose of simvastatin by 50% and limit dose to 20 mg daily | |
| Ranolazine | Lovastatin | Combination is acceptable to use if clinically indicated and an alternative non-CYP3A4 statin cannot be used. However, doses of lovastatin or simvastatin should not exceed 20 mg daily |
| Simvastatin | ||
| Ticagrelor | Atorvastatin | Combination is acceptable without dose limitations |
| Lovastatin | Limit dose of lovastatin to 40 mg daily | |
| Simvastatin | Limit dose of simvastatin to 40 mg daily | |
| Verapamil | Lovastatin | Limit dose of lovastatin to 20 mg daily |
| Simvastatin | Limit dose of simvastatin to 10 mg daily |
Modified with permission from Kellick et al. © 2014 Elsevier. Modified with permission from Wiggins et al. © 2016 American Heart Association, Inc. See additional references for prescribing information for each statin.
DDI indicates drug–drug interaction.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 27
Antiretroviral Therapy and Statin Drug Interactions
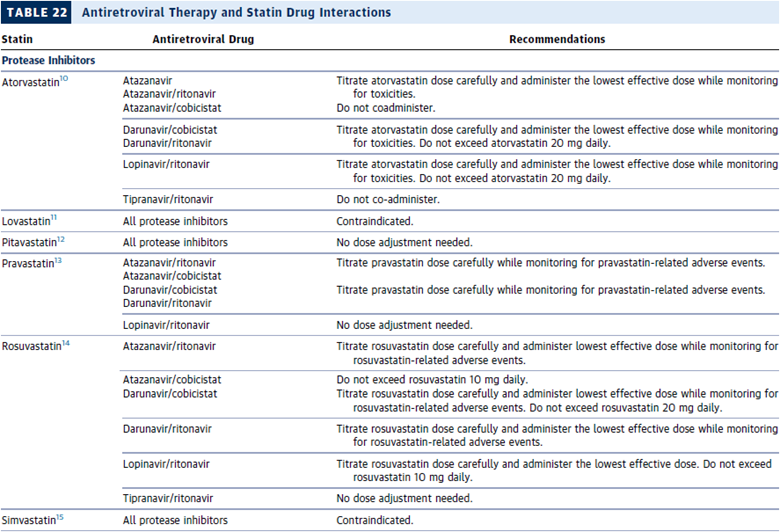
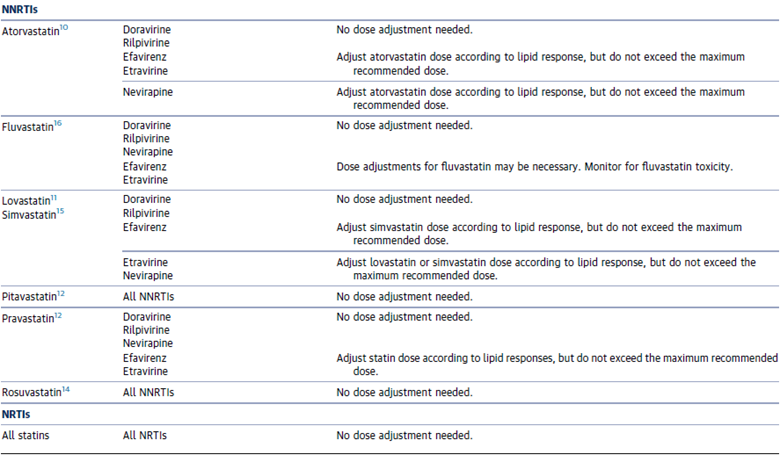
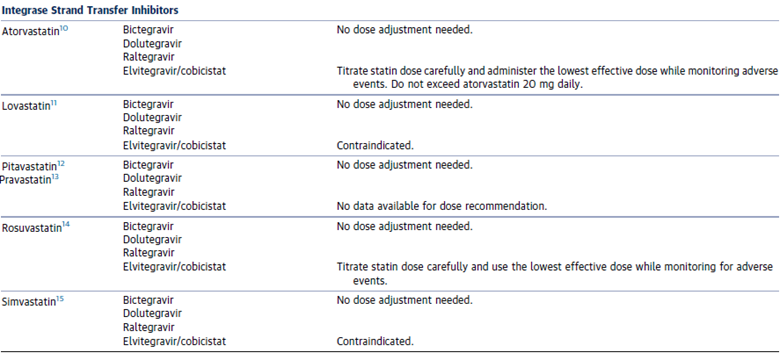
Adapted from and modified with permissions from Sarkar et al. Copyright 2000-2025 MDText.com, Inc.
NNRTI indicates non-nucleoside reverse transcriptase inhibitor; and NRTI, nucleoside reverse transcriptase inhibitor.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 22
Considerations Based on Ancestry
Considerations According to Ancestry Groups
Observed Higher Risk in Demographic Groups:
- South Asian ancestry is a high-risk demographic group and considered a risk enhancer.
- Increased prevalence of diabetes at lower BMI and waist circumferences across Asian populations.
- Increased CKM syndrome risk factors among Filipino American individuals, including hypertension, hyperlipidemia, and obesity.
Variation in Lipid Measures:
- Higher levels of Lp(a) are found in non-Hispanic Black individuals, but associated increase in CVD relative risk is similar to other groups.
- Elevated Lp(a) is associated with a higher population-attributed risk for MI among South Asian and Latin American people.
- Baseline CK levels are higher in non-Hispanic Black individuals but may not portend greater risk for statin-associated adverse events.
Statin Sensitivity:
- Some individuals of Chinese, Japanese, or Korean ancestry have been observed to have higher plasma concentrations of rosuvastatin, resulting in heightened sensitivity, and may benefit from starting rosuvastatin therapy at a lower dose.
- BMI indicates body mass index; CK, creatine kinase; CVD, cardiovascular disease; Lp(a), lipoprotein (a); and MI, myocardial infarction.
Source: 2026 ACC/AHA/Multisociety Guideline on the Management of Dyslipidemia, Table 21
Lifestyle Recommendations
The "2013 AHA/ACC Guideline on Lifestyle Management to Reduce Cardiovascular Risk" provides recommendations for heart-healthy lifestyle choices based on the latest research and evidence. The guidelines focus on two important lifestyle choices--diet and physical activity--which can have a drastic impact on cardiovascular health. Here's what every patient should know about the latest recommendations for reducing cardiovascular disease risk through diet and exercise.
Diet
- Diet is a vital tool for lowering cholesterol and blood pressure levels, which are two major risk factors for cardiovascular disease.
- Patients with high cholesterol and high blood pressure levels should eat plenty of vegetables, fruits and whole grains and incorporate low-fat dairy products, poultry, fish, legumes, non-tropical vegetable oils and nuts into their diet. They should also limit intake of sweets, sugar-sweetened beverages and red meats.
- There are many helpful strategies for heart-healthy eating, including the DASH diet and the USDA's Choose My Plate.
- Patients who need to lower their cholesterol should reduce saturated and trans fat intake. Ideally, only 5-6% of daily caloric intake should come from saturated fat.
- Patients with high blood pressure should consume no more than 2,400 mg of sodium a day, ideally reducing sodium intake to 1,500 mg a day. However, even reducing sodium intake in one's current diet by 1,000 mg each day can help lower blood pressure.
- It's important to adapt the recommendations above, keeping in mind calorie requirements, as well as, personal and cultural food preferences. Nutrition therapy for other conditions like diabetes should also be considered. Doing so helps create healthy eating patterns that are realistic and sustainable.
- Resources:
Source: www.CardioSmart.org/EatBetter
Physical Activity
- Regular physical activity helps lower cholesterol and blood pressure, reducing the risk for cardiovascular disease.
- In general, adults should engage in aerobic physical activity 3-4 times a week with each session lasting an average of 40 minutes.
- Moderate (brisk walking or jogging) to vigorous (running or biking) physical activity is recommended to reduce cholesterol levels.
- Resources:
Source: www.CardioSmart.org/MoveMore
The "2013 AHA/ACC/TOS Guideline for the Management of Overweight and Obesity in Adults" was created to reflect the latest research to outline best practices when it comes to treating obesity--a condition that affects more than one-third of American adults. These guidelines help address questions like "What's the best way to lose weight?" and "When is bariatric surgery appropriate?". Here is what every patient should know about the treatment of overweight and obesity:
- Definition of obesity: Obesity is a medical condition in which excess body fat has accumulated to the extent that it can have an adverse effect on one's health. Obesity can be diagnosed using body mass index (BMI), a measurement of height and weight, as well as waist circumference. Obesity is categorized as having a BMI of 30 or greater. Abdominal obesity is defined as having a waist circumference greater than 40 inches for a man or 35 inches for a woman.
- Benefits of weight loss: Obesity increases the risk for serious conditions such as cardiovascular disease, diabetes and death, but losing just a little bit of weight can result in significant health benefits. For an adult who is obese, losing just 3-5% of body weight can improve blood pressure and cholesterol levels and reduce the risk for cardiovascular disease and diabetes. Ideally, care providers recommend 5-10% weight loss for obese adults, which can produce even greater health benefits.
- Weight loss strategies: There is no single diet or weight loss program that works best for all patients. In general, reduced caloric intake and a comprehensive lifestyle intervention involving physical activity and behavior modification tailored according to a patient's preferences and health status is most successful for sustained weight loss. Further, weight loss interventions should include frequent visits with health care providers and last more than one year for sustained weight loss.
- Bariatric Surgery: Bariatric surgery may be a good option for severely obese patients to reduce their risk of health complications and improve overall health. However, bariatric surgery should be reserved for only the highest risk patients until more evidence is available on this issue. Present guidelines advise that weight loss surgery is only recommended for patients with extreme obesity (BMI ≥40) or in patients that have a BMI ≥35 in addition to a chronic health condition.
Resources:
Source: www.CardioSmart.org/Weight
Patient Resources
Fact Sheets, Infographics, and Action Plans:
Setting the Record Straight on Statins fact sheet
Statins are the gold standard for lowering cholesterol and reducing the risk of heart disease, heart attacks and stroke. They are a key part of treatment for people with existing heart disease too. But there is a lot of misinformation about statins. Here are some common statin myths, debunked.
https://www.cardiosmart.org/topics/high-cholesterol/assets/fact-sheet/setting-the-record-straight-on-statinsLower Your 'Bad' Cholesterol to Protect Your Heart infographic
Lowering your low-density lipoprotein (LDL), one type of fat in your blood, can lower your risk of heart attack, stroke and even death.
https://www.cardiosmart.org/topics/high-cholesterol/assets/infographic/lower-your-bad-cholesterol-to-protect-your-heartYour Action Plan for Lowering LDL Cholesterol and Heart Risks action plan
Use this workbook with your care team to come up with a plan to lower both your LDL cholesterol and risk of having a related heart attack or stroke.
https://www.cardiosmart.org/topics/high-cholesterol/assets/action-plan/your-action-plan-for-lowering-ldl-cholesterol-and-related-heart-risksUnderstanding Blood Lipids infographic
Cholesterol and triglycerides are types of fats in your blood. Having too much can clog your arteries and raise your risk of heart attack or stroke. Knowing your lipid test numbers is important.
https://www.cardiosmart.org/topics/high-cholesterol/assets/infographic/cholesterolWhat is Lipoprotein(a) or Lp(a)? infographic
Lp(a), pronounced "L-P little A" is like LDL cholesterol, but stickier, which can speed up narrowing of your arteries. If you have too much Lp(a), you have a higher chance of clogged arteries, heart attack, stroke, and heart valve problems.
https://www.cardiosmart.org/topics/high-cholesterol/assets/infographic/what-is-lipoproteina-or-lpaHigh Cholesterol Patient Hub
https://www.cardiosmart.org/topics/high-cholesterol
Stopping Smoking Condition Center
https://www.cardiosmart.org/topics/healthy-living/stop-smoking
Familial Hypercholesterolemia Patient Hub
https://www.cardiosmart.org/topics/familial-hypercholesterolemia
Clinician Resources - LDL-C Lowering Therapy
Understanding Cardiovascular Risk
10-Year ASCVD Risk
- The 10-year calculated ASCVD risk is a quantitative estimation of absolute risk based upon data from representative population samples.
- The 10-year risk estimate for "optimal risk factors" is represented by the following specific risk factor numbers for an individual of the same age, sex and race: Total cholesterol of ≤ 170 mg/dL, HDL-cholesterol of ≥ 50 mg/dL, untreated systolic blood pressure of ≤ 110 mm Hg, no diabetes history, and not a current smoker.
- While the risk estimate is applied to individuals, it is based on group averages.
- Just because two individuals have the same estimated risk does not mean that they will or will not have the same event of interest.
- Example: If the 10-year ASCVD risk estimate is 10%, this indicates that among 100 patients with the entered risk factor profile, 10 would be expected to have a heart attack or stroke in the next 10 years.
Lifetime ASCVD Risk
- The lifetime calculated ASCVD risk represents a quantitative estimation of absolute risk for a 50 year old man or woman with the same risk profile.
-
This estimation of risk is based on the
grouping of risk factor levels into 5 strata.
- All risk factors are optimal*
- ≥1 risk factors are not optimal†
- ≥1 risk factors are elevated‡
- 1 major risk factor§
- ≥2 major risk factors§
- The division of lifetime risk by these 5 strata leads to thresholds in the data with large apparent changes in lifetime risk estimates.
- Example: An individual that has all optimal risk factors except for a systolic blood pressure of 119 mm Hg has a lifetime ASCVD risk of 5%. In contrast, a similar individual that has all optimal risk factors except for a systolic blood pressure of 120 mm Hg has a lifetime ASCVD risk of 36%. This substantial difference in lifetime risk is due to the fact that they are in different stratum.
*Optimal risk levels for lifetime risk are represented by the simultaneous presence of all of the following: Untreated total cholesterol <180 mg/dL, untreated blood pressure <120/<80 mm Hg, no diabetes history, and not a current smoker
†Nonoptimal risk levels for lifetime risk are represented by 1 or more of the following: Untreated total cholesterol of 180 to 199 mg/dL, untreated systolic blood pressure of 120 to 139 mm Hg or diastolic blood pressure of 80 to 89 mm Hg, and no diabetes history and not a current smoker
‡Elevated risk levels for lifetime risk are represented by 1 or more of the following: Untreated total cholesterol of 200 to 239 mg/dL, untreated systolic blood pressure of 140 to 159 mm Hg or diastolic blood pressure of 90 to 99 mm Hg, and no diabetes history and not a current smoker
§Major risk levels for lifetime risk are represented by any of the following: Total cholesterol ≥240 mg/dL or treated, systolic blood pressure ≥160 mm Hg or diastolic blood pressure ≥100 mm Hg or treated, or diabetes, or current smoker
Lifestyle Recommendations
Diet recommendations
Diet recommendations for LDL-C lowering
-
Consume a dietary pattern that emphasizes
intake of vegetables, fruits, and whole
grains; includes low-fat dairy products,
poultry, fish, legumes, non-tropical vegetable
oils and nuts; and limits intake of sweets,
sugar-sweetened beverages, and red meats. (I
A)
- Adapt this dietary pattern to appropriate calorie requirements, personal and cultural food preferences, and nutrition therapy for other medical conditions (including diabetes mellitus).
- Achieve this pattern by following plans such as the DASH dietary pattern, the USDA Food Pattern, or the AHA Diet.
- Aim for a dietary pattern that achieves 5-6% of calories from saturated fat.
- Reduce percent of calories from saturated fat.
- Reduce percent of calories from trans fat. (I A)
Diet recommendations for blood pressure lowering
-
Consume a dietary pattern that emphasizes
intake of vegetables, fruits, and whole
grains; includes low-fat dairy products,
poultry, fish, legumes, non-tropical vegetable
oils and nuts; and limits intake of sweets,
sugar-sweetened beverages, and red meats. (I
A)
- Adapt this dietary pattern to appropriate calorie requirements, personal and cultural food preferences, and nutrition therapy for other medical conditions (including diabetes mellitus).
- Achieve this pattern by following plans such as the DASH dietary pattern, the USDA Food Pattern, or the AHA Diet.
- Lower sodium intake.
- Consume no more than 2400 mg of sodium per day. (I B)
Weight Management Recommendations
Diets for weight loss
-
Prescribe a diet to achieve reduced calorie
intake for obese or overweight individuals who
would benefit from weight loss, as part of a
comprehensive lifestyle intervention with 1 of
the following:
- 1200-1500 kcal/day for women and 1500-1800 kcal/day for men.
- 500-750 kcal/day energy deficit.
- Use one of the evidence-based diets that restricts certain food types (e.g., high-carbohydrate foods, low-fiber foods, or high-fat foods) in order to create an energy deficit by reduced food intake.
- Prescribe a calorie-restricted diet for obese or overweight individuals who would benefit from weight loss, based on the patient's preferences and health status, and preferably refer to a nutrition professional for counseling.
Lifestyle interventions and counseling for weight loss
- Advise participation in a comprehensive lifestyle program that assists participants in adhering to a lower calorie diet and increasing physical activity through the use of behavioral strategies.
- Prescribe on site, high-intensity (i.e., >14 sessions in 6 months) comprehensive weight loss interventions provided in individual or group sessions by a trained interventionist.
- Consider prescription of electronically delivered weight loss programs (including by telephone) that includes personalized feedback from a trained interventionist, recognizing that it may result in smaller weight loss than face-to-face interventions. (IIa A)
- Consider some commercial-based programs that provide comprehensive lifestyle interventions, provided there is peer-reviewed published evidence of their safety and efficacy. (IIa A)
- Consider a very low calorie diet (<800 kcal/day) only in limited circumstances and only when provided by trained practitioners in a medical care setting where medical monitoring and high intensity lifestyle intervention can be provided. (IIa A)
- Advise individuals who have lost weight to participate long term (>1 year) in a comprehensive weight loss maintenance program.
- Prescribe face-to-face or telephone-delivered weight loss maintenance programs that provide regular contact (> monthly) with a trained interventionist who helps participants engage in high levels of physical activity (i.e., 200-300 minutes/week), monitor body weight regularly (> weekly), and consume a reduced-calorie diet (need to lower body weight).
Selection criteria for bariatric surgical treatment of obesity
- Advise adults with a BMI ≥40 kg/m² or BMI ≥35 kg/m² with obesity-related co-morbid conditions who are motivated to lose weight and who have not responded to behavioral treatment with or without pharmacotherapy with sufficient weight loss to achieve targeted health outcome goals that bariatric surgery may be an appropriate option to improve health and offer referral to an experienced bariatric surgeon for consultation and evaluation. (IIa A)
Physical Activity Recommendations
Physical activity recommendations for modifying lipids and blood pressure lowering
-
Advise adults to engage in aerobic physical
activity to reduce LDL-cholesterol,
non-HDL-cholesterol, and blood pressure. (IIa
A)
- Frequency: 3-4 sessions a week
- Intensity: Moderate to vigorous
- Duration: 40 minutes on average
Physical activity recommendations for secondary prevention*
-
Aerobic exercise
- Frequency: 3-5 days/week
- Intensity: 50-80% of exercise capacity
- Duration: 20-60 minutes
- Modalities: Examples include walking, treadmill, cycling, rowing, stair climbing, and arm/leg ergometry
-
Resistance exercise
- Frequency: 2-3 days/week
- Intensity: 10-15 repetitions/set to moderate fatigue
- Duration: 1-3 sets of 8-10 upper and lower body exercises
- Modalities: Examples include calisthenics, elastic bands, cuff/hand weights, dumbbells, free weights, wall pulleys, and weight machines
Tobacco Cessation Recommendations
5 R's for patients not ready to quit
- Relevance—Encourage the patient to indicate why quitting is personally relevant.
- Risks—Ask the patient to identify potential negative consequences of tobacco use.
- Rewards—Ask the patient to identify potential benefits of stopping tobacco use.
- Roadblocks—Ask the patient to identify barriers or impediments to quitting.
- Repetition—The motivational intervention should be repeated every time an unmotivated patient has an interaction with a clinician. Tobacco users who have failed in previous quit attempts should be told that most people make repeated quit attempts before they are successful.
5 A's for patients that are ready to quit
- Ask—Systematically identify all tobacco users at every visit.
- Advise—Strongly urge all smokers to quit.
- Assess—Identify smokers willing to make a quit attempt.
- Assist—Aid the patient in quitting.
- Arrange—Schedule follow-up contact.
Groups that Benefit from Statin Therapy
1. Secondary Prevention: Clinical ASCVD
Clinical ASCVD includes acute coronary syndromes, history of MI, stable or unstable angina, coronary or other arterial revascularization, stroke, TIA, or peripheral arterial disease presumed to be of atherosclerotic origin.
High-intensity statin therapy should be initiated or continued as first-line therapy in women and men ≤75 years of age who have clinical ASCVD, unless contraindicated.
In individuals with clinical ASCVD in whom high-intensity statin therapy would otherwise be used, when high-intensity statin therapy is contraindicated or when characteristics predisposing to statin-associated adverse effects are present, moderate-intensity statin should be used as the second option if tolerated.
In individuals with clinical ASCVD >75 years of age, it is reasonable to evaluate the potential for ASCVD risk-reduction benefits and for adverse effects, drug-drug interactions and to consider patient preferences, when initiating a moderate- or high-intensity statin. It is reasonable to continue statin therapy in those who are tolerating it. (IIa B)
2. Primary Prevention: LDL-C ≥190 mg/dL
Individuals with LDL-C ≥190 mg/dL or triglycerides ≥500 mg/dL should be evaluated for secondary causes of hyperlipidemia. (I B)
Adults ≥21 years of age with primary LDL-C ≥190 mg/dL should be treated with high-intensity statin therapy unless contraindicated. For individuals unable to tolerate high-intensity statin therapy, use the maximum tolerated statin intensity. (I B)
For individuals ≥21 years of age with an untreated primary LDL-C ≥190 mg/dL, it is reasonable to intensify statin therapy to achieve at least a 50% LDL-C reduction. (IIa B)
For individuals ≥21 years of age with an untreated primary LDL-C ≥190 mg/dL, after the maximum intensity of statin therapy has been achieved, addition of a nonstatin drug may be considered to further lower LDL-C. Evaluate the potential for ASCVD risk reduction benefits, adverse effects, drug-drug interactions, and consider patient preferences. (IIb C)
3. Primary Prevention: Diabetes and aged 40 to 75 years with LDL-C between 70 - 189 mg/dL
Moderate-intensity statin therapy should be initiated or continued for adults 40 to 75 years of age with diabetes mellitus.
High-intensity statin therapy is reasonable for adults 40 to 75 years of age with diabetes mellitus with a ≥7.5% estimated 10-year ASCVD risk unless contraindicated. (IIa B)
In adults with diabetes mellitus, who are <40 or >75 years of age, it is reasonable to evaluate the potential for ASCVD benefits and for adverse effects, for drug-drug interactions, and to consider patient preferences when deciding to initiate, continue, or intensify statin therapy. (IIa C)
4. Primary Prevention: No diabetes and estimated 10-year ASCVD risk of ≥7.5% who are between 40 to 75 years of age with LDL-C between 70 - 189 mg/dL
The Pooled Cohort Equations should be used to estimate 10-year ASCVD risk for individuals with LDL-C 70 to 189 mg/dL without clinical ASCVD to guide initiation of statin therapy for the primary prevention of ASCVD. (I B)
Before initiating statin therapy for the primary prevention of ASCVD in adults with LDL-C 70 - 189 mg/dL without clinical ASCVD or diabetes it is reasonable for clinicians and patients to engage in a discussion which considers the potential for ASCVD risk reduction benefits and for adverse effects, for drug-drug interactions, and patient preferences for treatment. (IIa C)
Adults 40 to 75 years of age with LDL-C 70 to 189 mg/dL, without clinical ASCVD or diabetes and an estimated 10-year ASCVD risk ≥7.5% should be treated with moderate- to high-intensity statin therapy.
It is reasonable to offer treatment with a moderate-intensity statin to adults 40 to 75 years of age, with LDL-C 70 to 189 mg/dL, without clinical ASCVD or diabetes and an estimated 10-year ASCVD risk of 5% to <7.5%. (IIa B)
In adults with LDL-C <190 mg/dL who are not otherwise identified in a statin benefit group, or for whom after quantitative risk assessment a risk-based treatment decision is uncertain, additional factors may be considered to inform treatment decision making. In these individuals, statin therapy for primary prevention may be considered after evaluating the potential for ASCVD risk reduction benefits, adverse effects, drug-drug interactions, and discussion of patient preferences. (IIb C)
Additional Factors
These factors may include:
Statin benefit may be less clear in other groups; additional factors may be considered to inform treatment decision making.
- 5 to <7.5% 10-year ASCVD risk
- Primary LDL-C ≥160 mg/dL or other evidence of genetic hyperlipidemias
- Family history of premature ASCVD
- High sensitivity C-reactive protein ≥2 mg/L
- Coronary artery calcium score ≥300 Agatston units or ≥75th percentile for age, sex, and ethnicity
- Ankle-brachial index <0.9
- Lifetime risk of ASCVD
General Populations Recommendation Summary
| Primary Prevention Recommendations for Adults 40-75 Years LDL 70-189 mg/dL (1.7 - 4.8 mmol/L) | ||
|---|---|---|
| COR | LOE | Recommendations |
| I | A | 1. In adults at intermediate-risk, statin therapy reduces risk of ASCVD and in the context of a risk discussion, if a decision is made for statin therapy, a moderate- intensity statin should be recommended. |
| I | A | 2. In intermediate risk patients, LDL-C levels should be reduced by ≥ 30%, and for optimal ASCVD risk reduction, especially in high-risk patients, achieve LDL-C reductions of ≥ 50%. |
| I | B-NR | 3. For the primary prevention of clinical ASCVD* in adults 40 to 75 years of age without diabetes and with LDL-C 70 to 189 mg/dL (1.7 to 4.8 mmol/L), the 10-year ASCVD risk of a first "hard" ASCVD event (fatal and non-fatal MI or stroke) should be estimated using the race and sex-specific Pooled Cohort Equations (PCE) and adults should be categorized as low risk (<5%), borderline risk (5 to <7.5%), intermediate-risk (≥7.5 to <20%), and high-risk (≥20%). |
| I | B-NR | 4. Clinicians and patients should engage in a risk discussion that considers risk factors, adherence to healthy lifestyle, the potential for ASCVD risk-reduction benefits and the potential for adverse effects and drug–drug interactions, as well as patient preferences for an individualized treatment decision. |
| IIa | B-R | 5. In intermediate-risk adults, risk-enhancing factors favor initiation or intensification of statin therapy. |
| IIa | B-NR | 6. In intermediate-risk or selected borderline-risk adults, if the decision about statin usage remains uncertain, it is reasonable to use a coronary artery calcium (CAC) score in the decision to withhold, postpone or initiate statin therapy. |
| IIa | B-NR |
7. In intermediate-risk adults or selected
borderline-risk adults in whom a CAC score is
measured for the purpose of making a treatment
decision, AND
|
| IIb | B-R | 8. In intermediate-risk adults who would benefit from more aggressive LDL-C lowering and in whom high-intensity statins are advisable, but not acceptable or tolerated, it may be reasonable to add a non-statin drug (ezetimibe or bile acid sequestrant) to a moderate-intensity statin. |
| IIb | B-R | 9. In patients at borderline risk, in risk discussion, the presence of risk-enhancing factors may justify initiation of moderate-intensity statin therapy. |
| Recommenendations for Older Adults | ||
|---|---|---|
| COR | LOE | Recommendations |
| IIb | B-R | 1. In adults > 75 years of age with LDL-C of 70 to 189 mg/dL (1.7 to 4.8 mmol/L), initiating a moderate- intensity statin may be reasonable. |
| IIb | B-R | 2. In adults > 75 years of age, it may be reasonable to stop statin therapy when functional decline (physical or cognitive), multimorbidity, frailty or reduced life expectancy limit the potential benefits of statin therapy. |
| IIb | B-R | 3. In adults 76-80 years of age with LDL-C of 70 to 189 mg/dL (1.7 to 4.8 mmol/L), it may be reasonable to measure coronary artery calcium (CAC) to reclassify those with CAC = 0 to avoid statin therapy. |
| Recommenendations for Patients with Diabetes Mellitus | ||
|---|---|---|
| COR | LOE | Recommendations |
| I | A | 1. In adults 40 to 75 years of age with diabetes, regardless of estimated 10-year ASCVD risk, moderate-intensity statin therapy is indicated. |
| IIa | A | 2. In adults with diabetes who are 40 to 75 years of age and have LDL-C 70 to 189 mg/dL (1.7 to 4.8 mmol/L), it is reasonable to assess the 10-year risk of a first ASCVD event using the race and sex-specific Pooled Cohort Equations (PCE) to help stratify ASCVD risk. |
| IIa | B-NR | 3. In adults with diabetes who have multiple ASCVD risk factors, it is reasonable to prescribe high-intensity statin therapy with the aim to reduce LDL-C by ≥50%. |
| IIa | B-NR | 4. In adults with diabetes older than 75 years of age who are already on statin therapy, it is reasonable to continue statin therapy. |
| IIb | C-LD | 5. In adults with diabetes and 10-year ASCVD risk ≥20%, it may be reasonable to add ezetimibe to maximum tolerated statin therapy to reduce LDL-C by ≥50%. |
| IIb | C-LD | 6. In adults with diabetes older than 75 years, after a patient discussion of potential benefits and risks, it may be reasonable to initiate statin therapy. |
| IIb | C-LD | 7. In adults 20 to 39 years of age with diabetes either of long duration (≥10 years of Type 2 diabetes, ≥20 years of Type 1), and/or albuminuria (≥30 mcg albumin/mg creatinine), eGFR <60 ml/min/m², retinopathy, neuropathy, it may be reasonable to initiate statin therapy. |
| Recommenendations for Primary Severe Hypercholesterolemia (LDL-C ≥ 190 mg/dL (≥ 4.9 mmol/L)) | ||
|---|---|---|
| COR | LOE | Recommendations |
| I | B-R | 1. In patients 20 to 75 years of age with LDL-C ≥190 mg/dL (≥4.9 mmol/L), maximally-tolerated statin therapy is recommended. |
| IIa | B-R | 2. In patients 20 to 75 years of age with LDL-C ≥ 190 mg/dL (≥4.9 mmol/L), who achieve less than 50% reduction in LDL-C while receiving maximally-tolerated statin therapy, and/or have an LDL-C-≥100 mg/dL (≥2.6 mmol/L), ezetimibe therapy is reasonable. |
| IIb | B-R | 3. In patients 20 to 75 years of age with a baseline LDL-C ≥190 mg/dL (≥4.9 mmol/L), who achieve less than 50% reduction in LDL-C and have fasting triglycerides >300 mg/dL (>3.4 mmol/L) while taking maximally-tolerated statin and ezetimibe therapy, the addition of a bile acid sequestrant may be considered. |
| IIb | B-R | 4. In heterozygous familial hypercholesterolemia patients 30 to 75 years of age with LDL-C ≥100 mg/dL (≥2.6 mmol/L) while taking maximally-tolerated statin and ezetimibe therapy, the addition of a PCSK9 inhibitor may be considered. |
| IIb | C-LD | 5. In patients 40 to 75 years of age with a baseline LDL-C ≥220 mg/dL (≥5.7 mmol/L) who achieve on treatment LDL-C ≥130 mg/dL (≥3.4 mmol/L), while receiving maximally-tolerated statin and ezetimibe therapy, the addition of a PCSK9 inhibitor may be considered. |
| Value Statement: Uncertain Value (B-NR) | 6. Among patients with familial hypercholesterolemia without evidence of clinical ASCVD taking maximally-tolerated statin and ezetimibe therapy, PCSK9 inhibitors provide uncertain value at 2018 US list prices. | |
| Recommenendations for Statin Therapy Use in Patients with ASCVD | ||
|---|---|---|
| COR | LOE | Recommendations |
| I | A | 1. In patients ≤75 years of age with clinical ASCVD*, high-intensity statin therapy should be initiated or continued with the aim of achieving a ≥70% reduction in LDL-C. |
| I | A | 2. In patients with clinical ASCVD in whom high-intensity statin therapy is contraindicated or who experience statin-associated side effects, moderate-intensity statin therapy should be initiated or continued with the aim of achieving a 30-49% reduction in LDL-C. |
| IIa | B-R | 3. In patients with clinical ASCVD who are on maximally tolerated statin therapy and are judged to be at very high-risk, and have LDL-C ≥70 mg/dL (≥1.8 mmol/L), it is reasonable to add ezetimibe therapy. |
| IIa | A SR | 4. In patients with clinical ASCVD, who are judged to be very high-risk and who are on maximally tolerated statin therapy and ezetimibe and have LDL-C ≥70 mg/dL (≥1.8 mmol/L) or non-HDL-C ≥100 mg/dL (≥2.6 mmol/L), it is reasonable to add a PCSK9 inhibitor following a clinician-patient discussion about the net benefit, safety, and cost. |
| Value Statement: Low Value (LOE: B-NR) | 5. Among patients with clinical ASCVD at high-risk and taking maximally tolerated statin and ezetimibe therapy, PCSK9 inhibitors provide low economic value at 2018 US list prices. | |
| IIa | B-R | 6. In patients with clinical ASCVD older than 75 years, it is reasonable to initiate moderate or high-intensity statin therapy after evaluating the potential for ASCVD risk-reduction, adverse effects, drug-drug interactions, frailty, and patient preferences. |
| IIa | C-LD | 7. In patients with clinical ASCVD older than 75 years of age who are tolerating high-intensity statin therapy, it is reasonable to continue high-intensity statin therapy after evaluating the potential for ASCVD risk-reduction, adverse effects, drug-drug interactions, frailty, and patient preferences. |
| IIb | B-R | 8. In patients with clinical ASCVD who are receiving maximally tolerated statin therapy and LDL-C remains ≥70 mg/dL (≥1.8 mmol/L), it may be reasonable to add ezetimibe. |
| IIb | B-R | 9. In patients with heart failure with reduced ejection fraction due to ischemic heart disease who have a reasonable life expectancy (3-5 years) and are not already on a statin due to ASCVD, clinicians may consider initiation of moderate-intensity statin therapy to reduce the occurrence of ASCVD events. |
Recommendations for Initiation of Statin Therapy

This flow diagram is intended to serve as an easy reference guide summarizing recommendations for ASCVD risk assessment and treatment. Assessment of the potential for benefit and risk from statin therapy for ASCVD prevention provides the framework for clinical decision making incorporating patient preferences.
* Percent reduction in LDL–C can be used as an indication of response and adherence to therapy, but is not in itself a treatment goal.
† The Pooled Cohort Equations can be used to estimate 10-year ASCVD risk in individuals with and without diabetes. The estimator within this application should be used to inform decision making in primary prevention patients not on a statin.
‡ Consider moderate-intensity statin as more appropriate in low-risk individuals.
§ For those in whom a risk assessment is uncertain, consider factors such as primary LDL–C ≥160 mg/dL or other evidence of genetic hyperlipidemias, family history of premature ASCVD with onset <55 years of age in a first-degree male relative or <65 years of age in a first-degree female relative, hs-CRP >2 mg/L, CAC score ≥300 Agatston units, or ≥75th percentile for age, sex, and ethnicity (for additional information, see http://www.mesa-nhlbi.org/CACReference.aspx), ABI <0.9, or lifetime risk of ASCVD. Additional factors that may aid in individual risk assessment may be identified in the future.
|| Potential ASCVD risk-reduction benefits. The absolute reduction in ASCVD events from moderate- or high-intensity statin therapy can be approximated by multiplying the estimated 10-year ASCVD risk by the anticipated relative risk reduction from the intensity of statin initiated (~30% for moderate-intensity statin or ~45% for high-intensity statin therapy). The net ASCVD risk reduction benefit is estimated from the number of potential ASCVD events prevented with a statin compared to the number of potential excess adverse events.
¶ Potential adverse effects. The excess risk of diabetes is the main consideration in ~0.1 excess cases per 100 individuals treated with a moderate-intensity statin for 1 year and ~0.3 excess cases per 100 individuals treated with a high-intensity statin for 1 year. In RCTs, both statin-treated and placebo-treated participants experienced the same rate of muscle symptoms. The actual rate of statin-related muscle symptoms in the clinical population is unclear. Muscle symptoms attributed to statin therapy should be evaluated (see Statin Safety Recommendations).
ABI indicates ankle-brachial index; ASCVD, atherosclerotic cardiovascular disease; CAC, coronary artery calcium; hs-CRP, high-sensitivity C-reactive protein; LDL–C, low-density lipoprotein cholesterol; MI, myocardial infarction; RCT, randomized controlled trial.
Intensities of Statin Therapy
Low-Intensity
Daily dose lowers LDL-C, on average by approximately <30%
| dose | |
|---|---|
Moderate-Intensity
Daily dose lowers LDL-C, on average by approximately 30% to <50%
| dose | |
|---|---|
High-Intensity
Daily dose lowers LDL-C, on average by approximately ≥50%
| dose | |
|---|---|
Statins and doses that are approved by the U.S. FDA but were not tested in the RCTs reviewed are listed in parentheses
* Evidence from 1 RCT (down-titration if unable to tolerate atorvastatin 80 mg)
** Initiation of or titration to simvastatin 80 mg is not recommended by the FDA due to increased risk of myopathy, including rhabdomyolysis
Recommendations to Monitor Response to Statin Therapy

*Fasting lipid panel preferred. In a nonfasting individual, a non–HDL–C ≥ 220 mg/dL may indicate genetic hypercholesterolemia that requires further evaluation or a secondary etiology. If nonfasting triglycerides are ≥ 500 mg/dL, a fasting lipid panel is required.
†In those already on a statin, in whom baseline LDL–C is unknown, an LDL–C <100 mg/dL was observed in most individuals receiving high-intensity statin therapy in RCTs.
‡Refer to Statin Safety Recommendations
Statin Safety Recommendations
Statin Selection
To maximize the safety of statins, selection of the appropriate statin and dose in men and nonpregnant/nonnursing women should be based on patient characteristics, level of ASCVD risk, and potential for adverse effects.
Moderate-intensity statin therapy should be used in individuals in whom high-intensity statin therapy would otherwise be recommended when characteristics predisposing them to statin associated adverse effects are present.
Characteristics predisposing individuals to statin adverse effects include, but are not limited to: (I B)
- Multiple or serious comorbidities, including impaired renal or hepatic function.
- History of previous statin intolerance or muscle disorders.
- Unexplained ALT elevations >3 times ULN.
- Patient characteristics or concomitant use of drugs affecting statin metabolism.
- >75 years of age.
Additional characteristics that may modify the decision to use higher statin intensities may include, but are not limited to:
- History of hemorrhagic stroke.
- Asian ancestry.
Statin Dosage
- Decreasing the statin dose may be considered when 2 consecutive values of LDL-C levels are <40 mg/dL. (IIb C)
- It may be harmful to initiate simvastatin at 80 mg daily or increase the dose of simvastatin to 80 mg daily. (III A)
Creatine Kinase (CK)
- CK should not be routinely measured in individuals receiving statin therapy. (III A)
- Baseline measurement of CK is reasonable for individuals believed to be at increased risk for adverse muscle events based on a personal or family history of statin intolerance or muscle disease, clinical presentation, or concomitant drug therapy that might increase the risk for myopathy. (IIa C)
- During statin therapy, it is reasonable to measure CK in individuals with muscle symptoms, including pain, tenderness, stiffness, cramping, weakness, or generalized fatigue. (II C)
Muscle Symptoms
It is reasonable to evaluate and treat muscle symptoms, including pain, tenderness, stiffness, cramping, weakness, or fatigue, in statin-treated patients according to the following management algorithm: (IIa B)
- To avoid unnecessary discontinuation of statins, obtain a history of prior or current muscle symptoms to establish a baseline before initiating statin therapy.
- If unexplained severe muscle symptoms or fatigue develop during statin therapy, promptly discontinue the statin and address the possibility of rhabdomyolysis by evaluating CK, creatinine, and a urinalysis for myoglobinuria.
-
If mild to moderate muscle symptoms develop
during statin therapy:
- Discontinue the statin until the symptoms can be evaluated.
- Evaluate the patient for other conditions that might increase the risk for muscle symptoms (e.g., hypothyroidism, reduced renal or hepatic function, rheumatologic disorders such as polymyalgia rheumatica, steroid myopathy, vitamin D deficiency, or primary muscle diseases).
- If muscle symptoms resolve, and if no contraindication exists, give the patient the original or a lower dose of the same statin to establish a causal relationship between the muscle symptoms and statin therapy.
- If a causal relationship exists, discontinue the original statin. Once muscle symptoms resolve, use a low dose of a different statin.
- Once a low dose of a statin is tolerated, gradually increase the dose as tolerated.
- If, after 2 months without statin treatment, muscle symptoms or elevated CK levels do not resolve completely, consider other causes of muscle symptoms listed above.
- If persistent muscle symptoms are determined to arise from a condition unrelated to statin therapy, or if the predisposing condition has been treated, resume statin therapy at the original dose.
Hepatic Function
- Baseline measurement of hepatic transaminase levels (ALT) should be performed before initiating statin therapy. (I B)
- During statin therapy, it is reasonable to measure hepatic function if symptoms suggesting hepatotoxicity arise (e.g., unusual fatigue or weakness, loss of appetite, abdominal pain, dark colored urine or yellowing of the skin or sclera). (IIa C)
Diabetes
Individuals receiving statin therapy should be evaluated for new-onset diabetes mellitus according to the current diabetes screening guidelines. Those who develop diabetes mellitus during statin therapy should be encouraged to adhere to a heart healthy dietary pattern, engage in physical activity, achieve and maintain a healthy body weight, cease tobacco use, and continue statin therapy to reduce their risk of ASCVD events. (I B)
Age and Drug Regimen Consideration
For individuals taking any dose of statins, it is reasonable to use caution in individuals >75 years of age, as well as in individuals that are taking concomitant medications that alter drug metabolism, taking multiple drugs, or taking drugs for conditions that require complex medication regimens (e.g., those who have undergone solid organ transplantation or are receiving treatment for HIV). A review of the manufacturer's prescribing information may be useful before initiating any cholesterol-lowering drug. (IIa C)
Cognitive Impairment
For individuals presenting with a confusional state or memory impairment while on statin therapy, it may be reasonable to evaluate the patient for nonstatin causes, such as exposure to other drugs, as well as for systemic and neuropsychiatric causes, in addition to the possibility of adverse effects associated with statin drug therapy. (IIb C)
External Links & References
Patient Resources
Understanding My Cardiovascular Risk
The "2013 ACC/AHA Guideline on the Assessment of Cardiovascular Risk" provides clear recommendations for estimating cardiovascular disease risk. Risk assessments are extremely useful when it comes to reducing risk for cardiovascular disease because they help determine whether a patient is at high risk for cardiovascular disease, and if so, what can be done to address any cardiovascular risk factors a patient may have. Here are the highlights of the guideline:
-
Risk assessments are used to determine the likelihood of a patient developing cardiovascular disease, heart attack or stroke in the future. In general, patients at higher risk for cardiovascular disease require more intensive treatment to help prevent the development of cardiovascular disease.
-
Risk assessments are calculated using a number of factors including age, gender, race, cholesterol and blood pressure levels, diabetes and smoking status, and the use of blood pressure-lowering medications. Typically, these factors are used to estimate a patient's risk of developing cardiovascular disease in the next 10 years. For example, someone who is young with no risk factors for cardiovascular disease would have a very low 10-year risk for developing cardiovascular disease. However, someone who is older with risk factors like diabetes and high blood pressure will have a much higher risk of developing cardiovascular disease in the next 10 years.
-
If a preventive treatment plan is unclear based on the calculation of risk outlined above, care providers should take into account other factors such as family history and level of C-reactive protein. Taking this additional information into account should help inform a treatment plan to reduce a patient's 10-year risk of developing cardiovascular disease.
-
Calculating the 10-year risk for cardiovascular disease using traditional risk factors is recommended every 4-6 years in patients 20-79 years old who are free from cardiovascular disease. However, conducting a more detailed 10-year risk assessment every 4-6 years is reasonable in adults ages 40-79 who are free of cardiovascular disease. Assessing a patient's 30-year risk of developing cardiovascular disease can also be useful for patients 20-59 years of age who are free of cardiovascular disease and are not at high short-term risk for cardiovascular disease.
-
Risk estimations vary drastically by gender and race. Patients with the same traditional risk factors for cardiovascular disease such as high blood pressure can have a different 10-year risk for cardiovascular disease as a result of their sex and race.
-
After care providers and patients work together to conduct a risk assessment, it's important that they discuss the implications of their findings. Together, patients and their care providers should weigh the risks and benefits of various treatments and lifestyle changes to help reduce the risk of developing cardiovascular disease.
Source: www.cardiosmart.org
Diet and Physical Activity Recommendations
The "2013 AHA/ACC Guideline on Lifestyle Management to Reduce Cardiovascular Risk" provides recommendations for heart-healthy lifestyle choices based on the latest research and evidence. The guidelines focus on two important lifestyle choices--diet and physical activity--which can have a drastic impact on cardiovascular health. Here's what every patient should know about the latest recommendations for reducing cardiovascular disease risk through diet and exercise.
Diet
- Diet is a vital tool for lowering cholesterol and blood pressure levels, which are two major risk factors for cardiovascular disease.
- Patients with high cholesterol and high blood pressure levels should eat plenty of vegetables, fruits and whole grains and incorporate low-fat dairy products, poultry, fish, legumes, non-tropical vegetable oils and nuts into their diet. They should also limit intake of sweets, sugar-sweetened beverages and red meats.
- There are many helpful strategies for heart-healthy eating, including the DASH diet and the USDA's Choose My Plate.
- Patients who need to lower their cholesterol should reduce saturated and trans fat intake. Ideally, only 5-6% of daily caloric intake should come from saturated fat.
- Patients with high blood pressure should consume no more than 2,400 mg of sodium a day, ideally reducing sodium intake to 1,500 mg a day. However, even reducing sodium intake in one's current diet by 1,000 mg each day can help lower blood pressure.
- It's important to adapt the recommendations above, keeping in mind calorie requirements, as well as, personal and cultural food preferences. Nutrition therapy for other conditions like diabetes should also be considered. Doing so helps create healthy eating patterns that are realistic and sustainable.
Physical Activity
- Regular physical activity helps lower cholesterol and blood pressure, reducing the risk for cardiovascular disease.
- In general, adults should engage in aerobic physical activity 3-4 times a week with each session lasting an average of 40 minutes.
- Moderate (brisk walking or jogging) to vigorous (running or biking) physical activity is recommended to reduce cholesterol levels.
Source: www.cardiosmart.org
Weight Management Recommendations
The "2013 AHA/ACC/TOS Guideline for the Management of Overweight and Obesity in Adults" was created to reflect the latest research to outline best practices when it comes to treating obesity--a condition that affects more than one-third of American adults. These guidelines help address questions like "What's the best way to lose weight?" and "When is bariatric surgery appropriate?". Here is what every patient should know about the treatment of overweight and obesity:
-
Definition of obesity: Obesity is a medical condition in which excess body fat has accumulated to the extent that it can have an adverse effect on one's health. Obesity can be diagnosed using body mass index (BMI), a measurement of height and weight, as well as waist circumference. Obesity is categorized as having a BMI of 30 or greater. Abdominal obesity is defined as having a waist circumference greater than 40 inches for a man or 35 inches for a woman.
-
Benefits of weight loss: Obesity increases the risk for serious conditions such as cardiovascular disease, diabetes and death, but losing just a little bit of weight can result in significant health benefits. For an adult who is obese, losing just 3-5% of body weight can improve blood pressure and cholesterol levels and reduce the risk for cardiovascular disease and diabetes. Ideally, care providers recommend 5-10% weight loss for obese adults, which can produce even greater health benefits.
-
Weight loss strategies: There is no single diet or weight loss program that works best for all patients. In general, reduced caloric intake and a comprehensive lifestyle intervention involving physical activity and behavior modification tailored according to a patient's preferences and health status is most successful for sustained weight loss. Further, weight loss interventions should include frequent visits with health care providers and last more than one year for sustained weight loss.
-
Bariatric Surgery: Bariatric surgery may be a good option for severely obese patients to reduce their risk of health complications and improve overall health. However, bariatric surgery should be reserved for only the highest risk patients until more evidence is available on this issue. Present guidelines advise that weight loss surgery is only recommended for patients with extreme obesity (BMI ≥40) or in patients that have a BMI ≥35 in addition to a chronic health condition.
Source: www.cardiosmart.org
Blood Cholesterol Management Recommendations
The American College of Cardiology (ACC) and the American Heart Association (AHA) recently developed new standards for treating blood cholesterol. These recommendations are based on a thorough and careful review of the very latest, highest quality clinical trial research. They help care providers deliver the best care possible. This page provides some of the highlights from the new practice guidelines. The ultimate goal of the new cholesterol practice guidelines is to reduce a person's risk of heart attack, stroke and death. For this reason, the focus is not just on measuring and treating cholesterol, but identifying whether someone already has or is at risk for atherosclerotic cardiovascular disease (ASCVD) and could benefit from treatment.
What is ASCVD?
Heart attack and stroke are usually caused by atherosclerotic cardiovascular disease (ASCVD). ASCVD develops because of a build-up of sticky cholesterol-rich plaque. Over time, this plaque can harden and narrow the arteries.
These practice guidelines outline the most effective treatments that lower blood cholesterol in those individuals most likely to benefit. Most importantly, they were selected as the best strategies to lower cholesterol to help reduce future heart attack or stroke risk. Share this information with your health care provider so that you can ask questions and work together to decide what is right for you.
Key Points
Based on the most up-to-date and complete look at available clinical trial results:
-
Health care providers should focus on identifying those people who are most likely to have a heart attack or stroke and make sure they are given effective treatment to reduce their risk.
-
Cholesterol should be considered along with other factors known to make a heart attack or stroke more likely.
-
Knowing your risk of heart attack and stroke can help you and your health care provider decide whether you may need to take a medication—most likely a statin—to lower that risk.
-
If a medication is needed, statins are recommended as the first choice to lower heart attack and stroke risk among certain higher-risk patients based on an overwhelming amount of evidence. For those unable to take a statin, there are other cholesterol-lowering drugs; however, there is less research to support their use.
Evaluating Your Risk
Your health care provider will first want to assess your risk of ASCVD (assuming you don't already have it). This information will help determine if you are at high enough risk of a heart attack or stroke to need treatment.
To do this, your care provider will 1) review your medical history and 2) gauge your overall risk for heart attack or stroke. He/she will likely want to know:
-
whether you have had a heart attack, stroke or blockages in the arteries of your heart, neck, or legs.
-
your risk factors. In addition to your total cholesterol, LDL cholesterol, and HDL (so-called "good") cholesterol, your health care provider will consider your age, if you have diabetes, and whether you smoke and/or have high blood pressure.
-
about your lifestyle habits, other medical conditions, any previous drug treatments, and if anyone in your family has high cholesterol or suffered a heart attack or stroke at an early age.
A lipid or blood cholesterol panel will be needed as part of this evaluation. This blood test measures the amount of fatty substances (called lipids) in your blood. You may have to fast (not eat for a period of time) before having your blood drawn.
If there is any question about your risk of ASCVD, or whether you might benefit from drug therapy, your care provider may make additional assessments or order additional tests. The results of these tests can help you and your health care team decide what might be the best treatment for you. These tests may include:
-
Lifetime risk estimates —how likely you are to have a heart attack and stroke during your lifetime
-
Coronary artery calcium (CAC) score —a test that shows the presence of plaque or fatty build-up in the heart artery walls
-
High-sensitivity C-Reactive Protein (CRP) —a blood test that measures the amount of CRP, a marker of inflammation or irritation in the body; higher levels have been associated with heart attack and stroke
-
Ankle-brachial index (ABI) —the ratio of the blood pressure in the ankle compared to blood pressure in the arm, which can predict peripheral artery disease (PAD)
If you have very high levels of low-density lipoprotein (LDL or "bad") cholesterol, your care provider may want to find out if you have a genetic or familial form of hypercholesterolemia. This condition can be passed on in families.
Your Treatment Plan
Before coming up with a specific treatment plan, your care provider will talk with you about options for lowering your blood cholesterol and reducing your personal risk of atherosclerotic disease. This will likely include a discussion about heart-healthy living and whether you might benefit from a cholesterol-lowering medication.
Heart-Healthy Lifestyle
Adopting a heart-healthy lifestyle continues to be the first and best way to lower your risk of problems. Doing so can also help control or prevent other risk factors (for example: high blood pressure or diabetes). Experts suggest:
-
Eating a diet rich in vegetables, fruits, and whole grains ; this also includes low-fat dairy products, poultry, fish, legumes, and nuts; it limits intake of sweets, sugar-sweetened beverages and red meats.
-
Getting regular exercise ; check with your health care provider about how often and how much is right for you.
-
Maintaining a healthy weight .
-
Not smoking or getting help quitting .
-
Staying on top of your health , risk factors and medical appointments. For some people, lifestyle changes alone may not be enough to prevent a heart attack or stroke. In these cases, taking a statin at the right dose will most likely be necessary.
Medications
There are two types of cholesterol-lowering medications: statins and non-statins.
Statin Therapy
There is a large body of evidence that shows the use of a statin provides the greatest benefit and fewest safety issues. In particular, specific groups of patients appear to benefit most from taking moderate or high-intensity statin therapy. Based on this information, your care provider will likely recommend a statin if you have:
-
ASCVD
-
Very high LDL cholesterol (190 mg/dL or higher)
-
Type 2 diabetes and are between 40 and 75 years of age
-
Above a certain likelihood of having a heart attack or stroke in the next 10 years (7.5% or higher) and are between 40 and 75 years of age
In certain cases, your care provider may still recommend a statin even if you don't fit into one of the groups above. He/she will consider your overall health and other factors to help decide if you are at enough risk to benefit from a statin. Based on the guidelines, these may include:
-
Family history of premature heart attack or stroke
-
Your lifetime risk of ASCVD
-
LDL-cholesterol ≥160 mg/dL
-
hs-CRP ≥2 mg/L
-
Results from other special testing (CAC scoring, ABI)
If you are on a statin, your care provider will need to find the dose that is right for you.
-
People who have had a heart attack, stroke or other types of ASCVD tend to benefit the most from taking the highest amount (dose) of statin therapy if they tolerate it. This may be more appropriate than taking multiple drugs to lower cholesterol.
-
A more moderate dose of statin may be appropriate for some people with ASCVD, such as those over 75 years or those that might have problems taking the highest dose of a statin (i.e., those with prior organ transplantation).
Sometimes more than one statin needs to be tried before finding the one that works best.
If you are 75 years or older and have not already had a heart attack, stroke or other types of ASCVD, your care provider will discuss whether a statin is right for you.
Other cholesterol-lowering medications
Not all patients will be able to take the optimum dose of statin. After attention to lifestyle changes and statin therapy, non-statin drugs may be considered if you have high-risk with known ASCVD, diabetes, or very high LDL cholesterol values (≥190 mg/dL) and:
-
Have side effects from statins that prevent you from getting to the optimal dose or are not able to take a statin at all.
-
Are limited from taking an optimal dose due to other drugs that you are taking, including:
-
Transplant drug regimens to prevent rejection
-
Multiple drugs to treat HIV
-
Some antibiotics like erythromycin and clarithromycin or certain oral anti-fungal drugs
-
As always, it's important to talk with your health care provider about which medication is right for you.
What About Having Goals of Treatment?
Although keeping LDL-cholesterol lower with an optimal dose of statin is supported strongly by clinical trials, getting to a specific goal level is not.
Staying on Top of Your Risk
-
Take steps to lower your risk factors for heart attack, stroke and other problems —Make healthy choices (eating a healthy diet, getting exercise, maintaining a healthy weight and not smoking). Drug therapy, if needed, can help control risk factors.
-
Report side effects —Muscle aches are commonly reported and may or may not be due to the statin. If you are having problems, your care provider needs to know to help manage any side effects and possibly switch you to a different statin.
-
Take your medications as directed .
-
Get blood cholesterol and other tests that are recommended by your health care team. These can help assess whether statin therapy—and the dose—is working for you.
Questions to Ask
-
What are my risk factors for heart attack and stroke? Am I on the best prevention program to minimize this risk?
-
Is my cholesterol high enough that it might be due to a genetic condition?
-
What lifestyle changes can I make to stay healthy and prevent problems?
-
Do I need to be on a statin?
-
How do I monitor how I am doing?
-
What should I do if I develop muscle aches or weakness after starting the statin?
-
What do I do if I have other symptoms after starting the statin?
Source: www.cardiosmart.org
Common Cardiovascular Terms Alphabetical Glossary
For additional cardiovascular terms visit www.cardiosmart.org
Locked
The system has been locked
This version of the application has been
locked because of need to ugrade the science.
Please go to the store upgrade this application.



